Ophthalmic Drug CDMO Market Size, Share, Growth and Forecast (2026 - 2036)
Ophthalmic Drug CDMO Market is segmented by Product (Drug Product Manufacturing Services, Drug Development Services, Analytical & Regulatory Services, Packaging Services), Application (Eye Drops & Solutions, Ointments & Gels, Injectable Ophthalmics, Implants & Inserts), End Use (Pharmaceutical Companies, Biotechnology Companies, Research Institutes), and Region, with forecasts covering the period from 2026 to 2036.
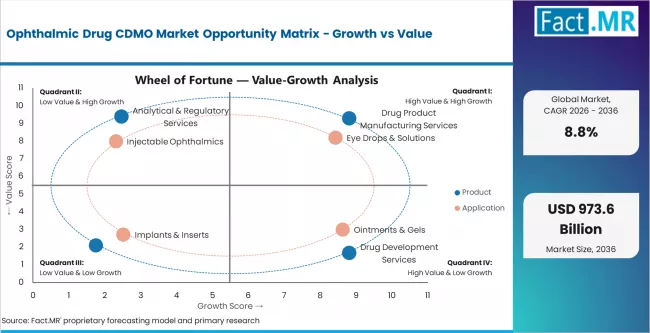
According to Fact.MR estimates, the global ophthalmic drug cdmo market market was valued at USD 385.0 billion in 2025. The market is projected to reach USD 418.9 billion in 2026 and is expected to grow to USD 973.6 billion by 2036, expanding at a CAGR of 8.8%. Drug Product Manufacturing Services is anticipated to account for 52.0% of the product segment in 2026, while Eye Drops & Solutions is expected to remain the leading application with around 46.0% share.
Ophthalmic Drug CDMO Market Analysis and Forecast by Fact.MR
According to Fact.MR estimates, the global ophthalmic drug cdmo market market was valued at USD 385.0 billion in 2025. The market is projected to reach USD 418.9 billion in 2026 and is expected to grow to USD 973.6 billion by 2036, expanding at a CAGR of 8.8%. Drug Product Manufacturing Services is anticipated to account for 52.0% of the product segment in 2026, while Eye Drops & Solutions is expected to remain the leading application with around 46.0% share.
Summary of Ophthalmic Drug CDMO Market
- Market Snapshot
- The ophthalmic drug cdmo market was valued at USD 384.96 million in 2025.
- By 2036, the Ophthalmic Drug CDMO Market market is expected to be worth USD 973.60 million.
- From 2026 to 2036, the market is projected to expand at a CAGR of 8.8%.
- The market is projected to create an incremental opportunity of USD 554.72 million between 2026 and 2036.
- In 2026, drug product manufacturing services are expected to account for 52% of the product segment, driven by sterile fill-finish and eye drop manufacturing demand.
- The USA (10.1%) and Germany (9.6%) are two of the fastest growing markets in the world.
- Demand and Growth Drivers
- Demand is shaped by expanding ophthalmic drug pipelines across glaucoma, dry eye disease, retinal disorders, and specialty ophthalmic indications from pharma and biotech developers.
- Growth reflects pharmaceutical company preference for specialty CDMO partners with sterile fill-finish capability and ophthalmic-specific formulation expertise.
- Adoption is increasing due to rising complexity of ophthalmic drug delivery including sustained release implants, preservative-free multi-dose systems, and intravitreal injection formulations.
- Product and Segment View
- Drug product manufacturing services lead the product segment with a 52% share in 2026, supported by sterile fill-finish and eye drop manufacturing capacity demand.
- Eye drops and solutions hold 46% of the application segment, anchored by glaucoma treatment and dry eye disease formulation volumes.
- Pharmaceutical companies account for 58% of end-use demand, while generic drug manufacturers, biotechnology companies, and ophthalmic biotech firms form secondary channels.
- Geography and Competitive Outlook
- North America remains the largest regional market, supported by strong ophthalmic pharma pipeline activity, mature CDMO partnership models, and established specialty manufacturing infrastructure.
- Europe follows closely, led by Germany where strong pharma manufacturing infrastructure and specialty ophthalmic drug development both support CDMO demand.
- The competitive field is led by Lonza Group with an estimated 33.9% share, followed by Catalent Inc., Recipharm AB, Siegfried Holding, and Piramal Pharma Solutions.
- Analyst Opinion
- The ophthalmic drug CDMO market represents a specialized pharmaceutical outsourcing segment where technical capability barriers protect premium margin economics, and the commercial positioning reflects this specialty character. The category is structurally attractive because ophthalmic drug formulations require sterile manufacturing capability, specialty primary packaging including multi-dose dropper systems, and formulation expertise spanning solutions, ointments, gels, and increasingly sustained-release implants. The commercial lever that matters most is integrated development and manufacturing capability because pharma customers increasingly prefer CDMO partners that can handle preformulation development, clinical trial material manufacturing, and commercial-scale production through single engagements. Players that combine deep sterile manufacturing capability with ophthalmic-specific formulation and packaging expertise will capture premium positioning, while generalist CDMO competitors without specialty ophthalmic capabilities face pressure as pharma customers consolidate ophthalmic work with specialist providers.
Why is Ophthalmic Drug CDMO Market Growing?
- Growth reflects expanding ophthalmic drug pipelines across glaucoma, dry eye disease, retinal disorders, and specialty ophthalmic indications.
- Demand is shaped by pharmaceutical company preference for specialty CDMO partners with sterile fill-finish capability and ophthalmic formulation expertise.
- Adoption is increasing due to rising complexity of ophthalmic drug delivery systems including sustained release implants and intravitreal injection formulations.
The demand base rests on expanding ophthalmic drug pipelines across multiple therapeutic categories. Glaucoma treatment remains the largest indication by volume, with ongoing formulation innovation around once-daily dosing, preservative-free options, and sustained-release drug delivery. Dry eye disease treatment has expanded substantially following new product approvals from companies such as Novartis, AbbVie, and specialty ophthalmic biotech firms. Retinal disease treatment pipelines including age-related macular degeneration, diabetic macular edema, and geographic atrophy continue producing new biologic and small-molecule product candidates requiring specialty CDMO capability.
The second structural driver is pharmaceutical company preference for specialty CDMO partners. Ophthalmic drug manufacturing requires specialty sterile fill-finish capability, expertise in ophthalmic-specific primary packaging including multi-dose dropper systems, and formulation expertise across solutions, ointments, and gels. Pharma companies increasingly prefer CDMO partners with dedicated ophthalmic capability rather than generalist sterile manufacturers, which concentrates demand at specialty providers such as Lonza Group, Catalent Inc., Recipharm AB, and other specialists.
The third driver is rising complexity of ophthalmic drug delivery. Sustained-release implants such as Durysta (AbbVie) and Yutiq, preservative-free multi-dose systems, and intravitreal injection formulations all require specialty manufacturing capability beyond traditional eye drop production. As ophthalmic drug delivery technology continues advancing, the technical capability bar for CDMO participation rises, which supports premium pricing for specialty providers with sustained investment in advanced formulation and manufacturing capabilities.
Segment-wise Analysis of Ophthalmic Drug CDMO Market
- Drug product manufacturing services lead the product segment with 52% share in 2026, anchored by sterile fill-finish and eye drop manufacturing volumes.
- Drug development services represent a fast-growing sub-segment, driven by pharma preference for integrated development and manufacturing engagements.
- Eye drops and solutions hold 46% of the application segment, followed by ointments and gels, injectable ophthalmics, and implants and inserts as secondary use cases.
The segmentation view shows a category where drug product manufacturing dominates volume while integrated development and manufacturing engagements drive premium growth. Competitive advantage rests on sterile fill-finish capability depth, ophthalmic-specific formulation expertise, and integrated development and manufacturing capability. Growth over the forecast period is expected to tilt toward injectable ophthalmics and implant and insert manufacturing as advanced drug delivery technology adoption expands.
Why Do Drug Product Manufacturing Services Lead the Product Segment?
Drug product manufacturing services lead the product segment because they represent the largest share of ophthalmic CDMO contract value. Sterile fill-finish services for eye drops, ointments, gels, and injectable ophthalmics all fall under this segment and drive the majority of recurring manufacturing revenue. Commercial-scale manufacturing of approved ophthalmic drugs generates sustained multi-year contracts that anchor CDMO revenue, while clinical trial material manufacturing contracts also contribute to drug product manufacturing service volume.
The segment leadership is reinforced by capacity investment economics. Specialty ophthalmic manufacturing facilities require substantial capital investment in sterile manufacturing capability, specialty primary packaging equipment, and regulatory-compliant quality systems. These investments create meaningful barriers to entry that protect existing specialty providers' pricing positions, which supports sustained segment leadership. Lonza Group, Catalent Inc., and other leading providers continue investing in sterile fill-finish capacity expansion to serve growing pharma demand.
Why are Drug Development Services a Fast-Growing Sub-Segment?
Drug development services are a fast-growing sub-segment because pharmaceutical and biotechnology companies increasingly prefer integrated development and manufacturing engagements with single CDMO partners. Preformulation and formulation development work for new ophthalmic drugs naturally leads to clinical trial material manufacturing and eventually commercial-scale production at the same facility, which supports customer retention and lifetime value expansion for CDMO providers.
Growth within drug development services is being lifted by ophthalmic biotech company activity. Specialty ophthalmic biotech firms typically lack in-house manufacturing capability and rely heavily on CDMO partners for both development and commercial manufacturing. As venture capital investment in ophthalmic biotech continues, development services contract volume expands in parallel. Rising complexity of ophthalmic drug delivery systems also increases the technical intensity of development services engagements, which supports premium pricing.
Key Growth Drivers, Constraints, and Market Scope
- Ophthalmic pipeline expansion and pharma CDMO preference for specialty partners are the structural demand foundations.
- Advanced drug delivery complexity and integrated development and manufacturing engagement growth are the key compositional shifts driving premium growth.
- Capacity constraints at specialty providers, regulatory complexity, and customer concentration remain the leading constraints.
The market scope is shaped by the combination of steady drug product manufacturing service demand and accelerating specialty development engagement growth. Competitive advantage sits in sterile fill-finish capability depth, ophthalmic-specific formulation expertise, and integrated development and manufacturing capability.
Growth Drivers
The primary driver is expanding ophthalmic drug pipelines across glaucoma, dry eye disease, retinal disorders, and specialty ophthalmic indications, which drives CDMO contract volume growth. A second driver is pharmaceutical company preference for specialty CDMO partners with sterile fill-finish capability and ophthalmic formulation expertise, which concentrates demand at specialty providers. A third driver is rising complexity of ophthalmic drug delivery systems including sustained release implants and intravitreal injection formulations, which supports premium pricing for advanced capability providers. These three forces combine to deliver a high single-digit growth trajectory with premium uplift from specialty delivery system manufacturing.
Constraints
The leading constraint is capacity constraint at specialty ophthalmic CDMO providers. Sterile fill-finish capacity is globally tight, which creates scheduling constraints and pricing power for specialty providers but also limits category growth potential. A second constraint is regulatory complexity. Ophthalmic drug manufacturing is subject to stringent FDA and EMA regulatory oversight, with facility inspections, quality system audits, and product-specific validation requirements creating ongoing compliance burden. A third constraint is customer concentration. Major pharmaceutical customers represent outsized share of CDMO revenue, which creates concentration risk and limits pricing leverage for providers against their largest customers.
Opportunities
The strongest opportunity lies in advanced drug delivery system manufacturing expansion, where premium pricing for specialty capability supports meaningful margin growth. A second opportunity is sterile fill-finish capacity expansion, where providers investing ahead of demand can capture market share as pharma customers seek reliable capacity. A third opportunity is emerging market development, particularly in India and South Korea, where specialty pharmaceutical manufacturing capability is expanding and local ophthalmic pharma activity is growing.
Regional Outlook Across Key Markets
.webp)
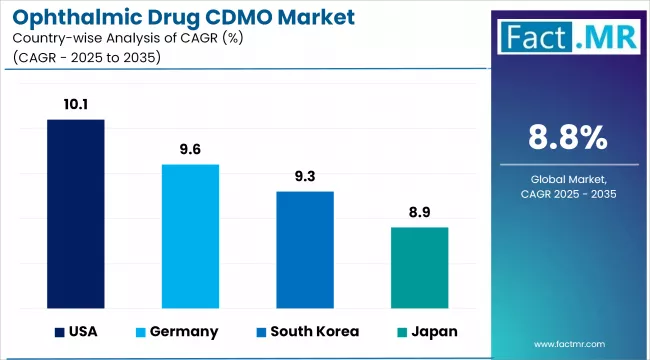
- The USA leads country-level growth at 10.1% CAGR, supported by strong ophthalmic pharma pipeline activity, mature CDMO partnership models, and established specialty manufacturing infrastructure.
- Germany follows at 9.6%, driven by strong pharma manufacturing infrastructure and specialty ophthalmic drug development activity.
- The UK at 9.3% and Japan at 8.9% both show active specialty pharmaceutical manufacturing and ophthalmic drug development programs.
- South Korea at 8.7% and France at 8.4% both support growing specialty CDMO positioning and ophthalmic pharma manufacturing capability.
Country CAGR Table
| Country | CAGR (%) |
|---|---|
| USA | 10.10% |
| Germany | 9.60% |
| UK | 9.30% |
| Japan | 8.90% |
| South Korea | 8.70% |
| France | 8.40% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
Regional dynamics reflect the concentration of specialty ophthalmic CDMO activity in developed markets with strong pharmaceutical manufacturing infrastructure. The USA leads on the strength of ophthalmic pharma pipeline depth and mature CDMO partnership models. Europe, led by Germany, supports active specialty CDMO activity through established pharmaceutical manufacturing infrastructure. Asia Pacific growth is led by Japan and South Korea, where specialty pharmaceutical manufacturing capability continues expanding.
United States
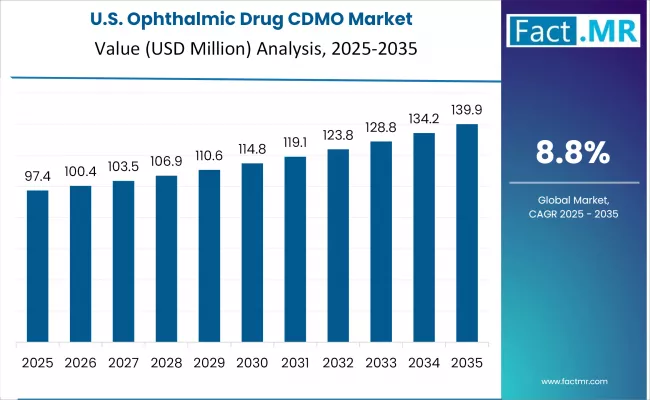
The United States anchors global demand through its strong ophthalmic pharma pipeline activity, mature CDMO partnership models, and established specialty manufacturing infrastructure. AbbVie (Allergan), Novartis, Alcon, Bausch & Lomb, and specialty ophthalmic pharma and biotech companies drive substantial CDMO contract volume. Lonza Group, Catalent Inc., Thermo Fisher Scientific, and Patheon all operate US specialty ophthalmic manufacturing capacity serving domestic and export markets.
- Strong ophthalmic pharma pipeline activity drives sustained CDMO contract volume.
- Mature CDMO partnership models support integrated development and manufacturing engagements.
- Established specialty manufacturing infrastructure supports premium capability positioning.
Germany
Germany leads European specialty CDMO activity on the strength of mature pharmaceutical manufacturing infrastructure, strong specialty ophthalmic drug development activity, and established CDMO service provider presence. Recipharm AB and other European specialty CDMO providers maintain significant German manufacturing capacity. German pharmaceutical manufacturers including Bayer support domestic ophthalmic pipeline activity, while specialty biotech firms also drive CDMO demand.
- Mature pharmaceutical manufacturing infrastructure supports specialty CDMO capacity.
- Strong specialty ophthalmic drug development supports CDMO contract volume.
- Established CDMO service provider presence anchors regional capability.
United Kingdom
The United Kingdom market is shaped by active specialty pharmaceutical manufacturing, strong ophthalmic biotech activity particularly around Cambridge and Oxford clusters, and established CDMO service provider presence. GSK supports domestic ophthalmic pipeline activity, while specialty biotech firms drive additional CDMO contract volume. Almac Group and other UK-based specialty providers serve domestic and export demand.
- Active specialty pharmaceutical manufacturing supports CDMO capability positioning.
- Strong ophthalmic biotech activity in Cambridge and Oxford clusters drives contract volume.
- Established CDMO service provider presence anchors specialty capability.
Japan
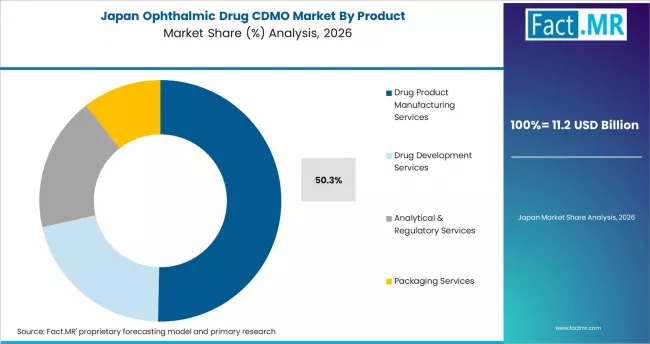
Japan represents a mature specialty CDMO market shaped by strong domestic ophthalmic pharma activity including Santen Pharmaceutical and Otsuka Pharmaceutical, established specialty manufacturing infrastructure, and PMDA regulatory oversight. Santen is the global leader in ophthalmic pharmaceuticals and drives substantial specialty manufacturing demand. Japanese CDMO providers also serve export demand to other Asia Pacific markets.
- Strong domestic ophthalmic pharma activity led by Santen supports specialty CDMO demand.
- Established specialty manufacturing infrastructure supports domestic and export capability.
- PMDA regulatory oversight supports high-quality specialty manufacturing standards.
South Korea
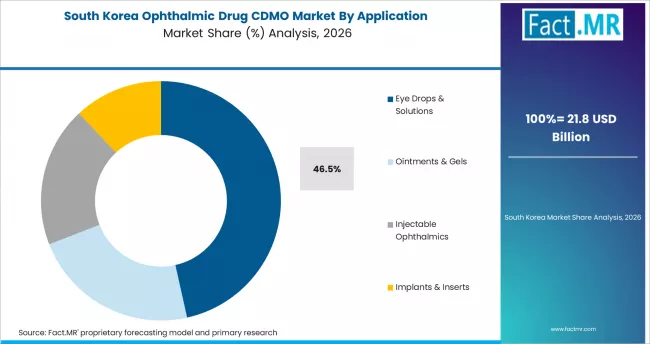
South Korea represents a growing specialty CDMO market driven by expanding domestic pharmaceutical manufacturing capability, rising ophthalmic biotech activity, and cross-border CDMO partnerships with global pharma. Korean CDMO providers are building specialty ophthalmic capability to serve both domestic pharma customers and export opportunities particularly to other Asia Pacific markets.
- Expanding domestic pharmaceutical manufacturing supports specialty CDMO capability.
- Rising ophthalmic biotech activity drives domestic contract volume growth.
- Cross-border CDMO partnerships support export opportunity development.
France
France represents a steady specialty CDMO market shaped by Sanofi's domestic pharmaceutical activity, specialty ophthalmic biotech firms, and established CDMO service provider presence. Corden Pharma and other French-based specialty providers serve domestic and European pharma customers. French ophthalmic biotech activity supports additional CDMO contract volume.
- Sanofi's domestic pharmaceutical activity supports specialty CDMO demand.
- Specialty ophthalmic biotech firms drive additional contract volume.
- Established CDMO service provider presence anchors regional specialty capability.
Competitive Benchmarking and Company Positioning
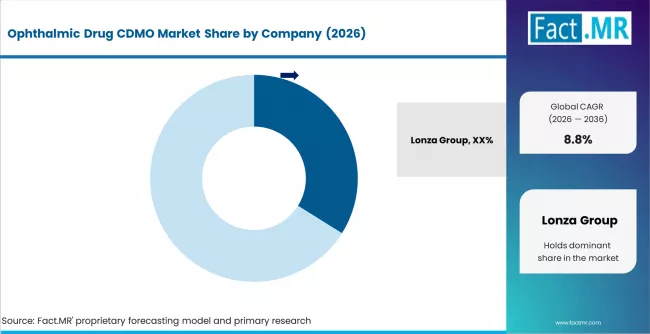
- Lonza Group leads with an estimated 33.9% share, supported by its broad specialty CDMO portfolio, global manufacturing footprint, and integrated development and manufacturing capability.
- Catalent Inc., Recipharm AB, Siegfried Holding, and Piramal Pharma Solutions compete through differentiated specialty manufacturing capabilities and channel strength.
- Specialty players such as Thermo Fisher Scientific, Evonik Industries, Patheon, Almac Group, and Corden Pharma hold focused positions across specific service segments.
The competitive field is concentrated at the top, with Lonza Group commanding a strong share position through its broad specialty CDMO portfolio spanning development services, sterile fill-finish manufacturing, and specialty formulation capabilities. Lonza's scale reach, global manufacturing footprint, and integrated development and manufacturing capability create meaningful competitive advantages across ophthalmic CDMO engagements.
Catalent Inc. competes through its broad pharma and biotech CDMO services including specialty ophthalmic capability. Recipharm AB holds strong positions in European specialty manufacturing. Siegfried Holding and Piramal Pharma Solutions both maintain specialty ophthalmic capacity serving pharma customers globally. Thermo Fisher Scientific and Patheon compete through broader CDMO portfolios with ophthalmic capability. Evonik Industries holds specialty positions in drug delivery platform services. Almac Group, Corden Pharma, and other specialty providers maintain focused positions in specific service segments.
Strategic priorities across the field include expanding sterile fill-finish capacity, deepening ophthalmic-specific formulation expertise, and building advanced drug delivery system manufacturing capability. Scaled specialty providers with integrated development and manufacturing capability will capture the bulk of forecast period growth, while generalist CDMO competitors without specialty ophthalmic depth face pressure as pharma customers consolidate ophthalmic work with specialist partners.
Competitive Analysis
Competitive positioning rests on three dimensions: sterile fill-finish capability depth, ophthalmic-specific formulation expertise, and integrated development and manufacturing capability. Lonza Group leads across all three dimensions. Catalent Inc. competes broadly through portfolio depth. Recipharm AB and Siegfried Holding lead specialty manufacturing positioning in European markets. Evonik Industries leads specifically in drug delivery platform capability.
Second-tier competitors maintain relevance through specialty service positioning, focused therapeutic strategies, or regional channel concentration. Indian and Korean specialty CDMO providers are building capability positions in value-tier segments, particularly for generic ophthalmic drug manufacturing. The gap between integrated specialty providers with development and manufacturing capability and single-service competitors continues to widen as pharma customers consolidate CDMO partnerships.
Benchmarking Table
| Company | Scale Reach | Fill-Finish Capacity | Ophthalmic Expertise | Development Services |
|---|---|---|---|---|
| Lonza Group | Global | Strong | Strong | Strong |
| Catalent Inc. | Global | Strong | Medium-Strong | Strong |
| Recipharm AB | Global | Medium-Strong | Medium-Strong | Medium |
| Siegfried Holding | Global | Medium-Strong | Medium | Medium |
| Piramal Pharma Solutions | Global | Medium | Medium | Medium |
| Thermo Fisher Scientific | Global | Strong | Medium | Strong |
| Evonik Industries | Global | Medium | Medium-Strong | Medium-Strong |
Recent Developments
- In 2025, Lonza Group continued expanding its specialty CDMO portfolio through capacity investments targeting sterile fill-finish and advanced drug delivery system manufacturing.
- In 2025, Catalent Inc. advanced its specialty CDMO capability through platform updates and integration with pharma customer development pipelines.
- In 2026, Recipharm AB strengthened its European specialty manufacturing capacity through targeted expansion investments supporting ophthalmic and sterile manufacturing applications.
Leading Companies Shaping Ophthalmic Drug CDMO Market
-
Major Players
- Lonza Group
- Catalent Inc.
- Recipharm AB
- Siegfried Holding
- Piramal Pharma Solutions
- Thermo Fisher Scientific
- Evonik Industries
- Patheon
-
Emerging Players
- Almac Group
- Corden Pharma
- Vetter Pharma
- Baxter BioPharma Solutions
- Pfizer CentreOne
- WuXi AppTec
- Samsung Biologics
- Fareva Group
Sources and Research References
- United States Food and Drug Administration (FDA) - NDA and BLA Filings for Ophthalmic Drugs
- European Medicines Agency (EMA) - Centralized Procedure Filings for Ophthalmic Products
- World Health Organization (WHO) - Essential Medicines List Ophthalmic Publications
- American Academy of Ophthalmology (AAO) - Preferred Practice Pattern Publications
- Lonza Group - Annual Report and CDMO Segment Disclosures
- Catalent Inc. - Specialty CDMO Portfolio Publications
- Primary interviews with pharma company CDMO managers, biotech company operations leaders, CDMO business development teams, and regulatory specialists
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the current and future size of the ophthalmic drug CDMO market?
- How fast is the market expected to grow between 2026 and 2036?
- Which product segment is likely to lead the market by 2026?
- Which application segment will account for the highest demand by 2026?
- How is ophthalmic drug pipeline activity reshaping CDMO demand patterns?
- Why are the USA and Germany the fastest-growing markets?
- Which countries are projected to show the fastest growth through 2036?
- Who are the key companies active in the ophthalmic drug CDMO market?
- How is advanced drug delivery technology changing the competitive dynamics?
Ophthalmic Drug CDMO Market Definition
The ophthalmic drug CDMO market covers contract development and manufacturing services provided to pharmaceutical and biotechnology companies for ophthalmic drug products. This includes drug product manufacturing services such as sterile fill-finish and eye drop and solution manufacturing, drug development services including preformulation and formulation development and clinical trial material manufacturing, analytical and regulatory services including stability testing and regulatory filing support, and packaging services including ophthalmic bottle packaging and multi-dose and preservative-free systems. The market spans pharmaceutical companies, large pharma companies, generic drug manufacturers, biotechnology companies, ophthalmic biotech firms, gene therapy developers, research institutes, and clinical research organizations across product applications including eye drops and solutions, ointments and gels, injectable ophthalmics, and implants and inserts.
Ophthalmic Drug CDMO Market Inclusions
The scope includes drug product manufacturing services across sterile fill-finish services, eye drop and solution manufacturing, and ointment and gel formulation, drug development services including preformulation and formulation development and clinical trial material manufacturing, analytical and regulatory services including stability testing services and regulatory filing and compliance support, and packaging services including ophthalmic bottle packaging and multi-dose and preservative-free systems. It covers applications across eye drops and solutions for glaucoma treatment drops and dry eye disease formulations, ointments and gels including anti-infective ointments and anti-inflammatory gels, injectable ophthalmics including intravitreal injections and retinal disease treatments, and implants and inserts including sustained release implants and intraocular drug delivery systems. Coverage spans pharmaceutical companies, large pharma companies, generic drug manufacturers, biotechnology companies, ophthalmic biotech firms, gene therapy developers, research institutes, clinical research organizations, and academic research centers from 2026 to 2036.
Ophthalmic Drug CDMO Market Exclusions
The scope excludes in-house ophthalmic drug manufacturing performed by pharmaceutical companies at their own facilities without CDMO engagement. It also excludes active pharmaceutical ingredient (API) manufacturing services, which represent a separate CDMO segment with different technical and commercial dynamics. Ophthalmic medical device manufacturing services including intraocular lens production, contact lens manufacturing, and surgical instrument production are outside the scope. Diagnostic ophthalmic product development and manufacturing are also excluded, as are consumer ophthalmic products such as over-the-counter eye drops manufactured for retail channels rather than under pharma CDMO engagements.
Ophthalmic Drug CDMO Market Research Methodology
- Primary Research: Interviews with pharma company CDMO managers, biotech company operations leaders, CDMO business development teams, and regulatory specialists to validate contract structures, pricing dynamics, and capability requirements.
- Desk Research: Review of FDA NDA and BLA filings for ophthalmic drugs, CDMO company disclosures, PharmaCompass CDMO database, and published ophthalmic pharmaceutical pipeline publications.
- Market Sizing: Demand-side model built on ophthalmic drug approvals and pipeline activity, CDMO contract value estimates, per-project pricing across development and commercial manufacturing services, and capacity utilization patterns at specialty ophthalmic CDMO facilities.
- Data Validation: Cross-validation across primary interviews, CDMO financial disclosures, pharma company outsourcing spend analyses, and specialty pharmaceutical industry publications, with sensitivity analysis on ophthalmic pipeline approval rates and sterile fill-finish capacity investment.
Scope of Analysis
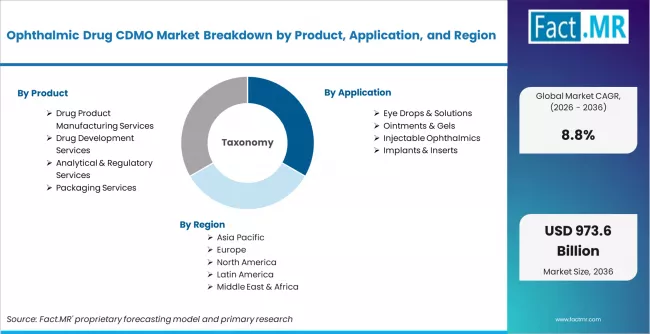
| Parameter | Details |
|---|---|
| Quantitative Units | USD 418.88 million to USD 973.60 million, at a CAGR of 8.8% |
| Market Definition | The ophthalmic drug CDMO market covers contract development and manufacturing services provided to pharmaceutical and biotechnology companies for ophthalmic drug products, spanning drug product manufacturing, drug development services, analytical and regulatory services, and packaging services across eye drops and solutions, ointments and gels, injectable ophthalmics, and implants and inserts. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, Germany, UK, Japan, South Korea, France, 30 plus countries |
| Key Companies | Lonza Group, Catalent Inc., Recipharm AB, Siegfried Holding, Piramal Pharma Solutions, Thermo Fisher Scientific, Evonik Industries, Patheon |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on ophthalmic drug approvals and pipeline activity, CDMO contract value estimates, per-project pricing across development and commercial manufacturing services, capacity utilization patterns at specialty ophthalmic CDMO facilities, and primary interviews across pharma company CDMO managers, biotech company operations leaders, and CDMO business development teams |
Market Segmentation Analysis
-
Ophthalmic Drug CDMO Market Market Segmented by Product:
- Drug Product Manufacturing Services
- Sterile Fill-Finish Services
- Eye Drop & Solution Manufacturing
- Ointment & Gel Formulation
- Sterile Fill-Finish Services
- Drug Development Services
- Preformulation & Formulation Development
- Clinical Trial Material Manufacturing
- Preformulation & Formulation Development
- Analytical & Regulatory Services
- Stability Testing Services
- Regulatory Filing & Compliance Support
- Stability Testing Services
- Packaging Services
- Ophthalmic Bottle Packaging
- Multi-Dose & Preservative-Free Systems
- Ophthalmic Bottle Packaging
- Drug Product Manufacturing Services
-
Ophthalmic Drug CDMO Market Market Segmented by Application:
- Eye Drops & Solutions
- Glaucoma Treatment Drops
- Dry Eye Disease Formulations
- Glaucoma Treatment Drops
- Ointments & Gels
- Anti-Infective Ointments
- Anti-Inflammatory Gels
- Anti-Infective Ointments
- Injectable Ophthalmics
- Intravitreal Injections
- Retinal Disease Treatments
- Intravitreal Injections
- Implants & Inserts
- Sustained Release Implants
- Intraocular Drug Delivery Systems
- Sustained Release Implants
- Eye Drops & Solutions
-
Ophthalmic Drug CDMO Market Market Segmented by End Use:
- Pharmaceutical Companies
- Large Pharma Companies
- Generic Drug Manufacturers
- Large Pharma Companies
- Biotechnology Companies
- Ophthalmic Biotech Firms
- Gene Therapy Developers
- Ophthalmic Biotech Firms
- Research Institutes
- Clinical Research Organizations
- Academic Research Centers
- Clinical Research Organizations
- Pharmaceutical Companies
-
Ophthalmic Drug CDMO Market Market Segmented by Technology:
- Sterile Manufacturing Technology
- Aseptic Fill-Finish Systems
- Contamination Control Systems
- Aseptic Fill-Finish Systems
- Controlled Drug Delivery Technology
- Sustained Release Formulations
- Nanoparticle-Based Delivery
- Sustained Release Formulations
- Advanced Formulation Technology
- Emulsions & Suspensions
- Preservative-Free Formulations
- Emulsions & Suspensions
- Analytical & Quality Control Technology
- Bioavailability Testing
- Stability & Sterility Testing
- Bioavailability Testing
- Sterile Manufacturing Technology
-
Ophthalmic Drug CDMO Market Market Segmented by Distribution Channel:
- Direct CDMO Contracts
- Pharma-CDMO Partnerships
- Long-Term Manufacturing Agreements
- Pharma-CDMO Partnerships
- Outsourcing Platforms
- Contract Manufacturing Networks
- Integrated Development Platforms
- Contract Manufacturing Networks
- Third-Party Service Providers
- Consulting & Regulatory Firms
- Clinical Trial Supply Vendors
- Consulting & Regulatory Firms
- Direct CDMO Contracts
-
Ophthalmic Drug CDMO Market Market Segmented by Packaging:
- Primary Packaging
- Ophthalmic Bottles
- Multi-Dose Dropper Systems
- Unit Dose Packaging
- Ophthalmic Bottles
- Secondary Packaging
- Cartons & Protective Packaging
- Cold Chain Packaging
- Cartons & Protective Packaging
- Primary Packaging
-
Ophthalmic Drug CDMO Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the ophthalmic drug CDMO market in 2026?
The global ophthalmic drug CDMO market is estimated to be valued at USD 418.88 million in 2026.
What will be the size of the ophthalmic drug CDMO market in 2036?
The market size is projected to reach USD 973.60 million by 2036.
How much will the ophthalmic drug CDMO market grow between 2026 and 2036?
The market is expected to grow at an 8.8% CAGR between 2026 and 2036.
Which product segment is expected to lead the market in 2026?
Drug product manufacturing services are expected to lead the product segment with a 52% share in 2026.
Which country is the fastest-growing market?
The USA is the fastest-growing country in the forecast, projected to grow at a 10.1% CAGR through 2036.
Why is Germany a key growth market?
Germany is projected to grow at a 9.6% CAGR, driven by strong pharma manufacturing infrastructure and specialty ophthalmic drug development activity.
What does the ophthalmic drug CDMO market cover?
The market covers contract development and manufacturing services provided to pharmaceutical and biotechnology companies for ophthalmic drug products, spanning drug product manufacturing, drug development services, analytical and regulatory services, and packaging services across eye drops and solutions, ointments and gels, injectable ophthalmics, and implants and inserts.
How is the market forecast estimated and validated?
The forecast is built on a hybrid demand-side and top-down model using ophthalmic drug approvals and pipeline activity, CDMO contract value estimates, per-project pricing across development and commercial manufacturing services, capacity utilization patterns at specialty ophthalmic CDMO facilities, and primary interviews across pharma company CDMO managers, biotech company operations leaders, and CDMO business development teams.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Drug Product Manufacturing Services
- Sterile Fill-Finish Services
- _Eye Drop & Solution Manufacturing
- _Ointment & Gel Formulation
- Drug Development Services
- Preformulation & Formulation Development
- _Clinical Trial Material Manufacturing
- Analytical & Regulatory Services
- Stability Testing Services
- _Regulatory Filing & Compliance Support
- Packaging Services
- Ophthalmic Bottle Packaging
- _Multi-Dose & Preservative-Free Systems
- Drug Product Manufacturing Services
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Eye Drops & Solutions
- Glaucoma Treatment Drops
- _Dry Eye Disease Formulations
- Ointments & Gels
- Anti-Infective Ointments
- _Anti-Inflammatory Gels
- Injectable Ophthalmics
- Intravitreal Injections
- _Retinal Disease Treatments
- Implants & Inserts
- Sustained Release Implants
- _Intraocular Drug Delivery Systems
- Eye Drops & Solutions
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical Companies
- Large Pharma Companies
- _Generic Drug Manufacturers
- Biotechnology Companies
- Ophthalmic Biotech Firms
- _Gene Therapy Developers
- Research Institutes
- Clinical Research Organizations
- _Academic Research Centers
- Pharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Sterile Manufacturing Technology
- Aseptic Fill-Finish Systems
- _Contamination Control Systems
- Controlled Drug Delivery Technology
- Sustained Release Formulations
- _Nanoparticle-Based Delivery
- Advanced Formulation Technology
- Emulsions & Suspensions
- _Preservative-Free Formulations
- Analytical & Quality Control Technology
- Bioavailability Testing
- _Stability & Sterility Testing
- Sterile Manufacturing Technology
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct CDMO Contracts
- Pharma-CDMO Partnerships
- _Long-Term Manufacturing Agreements
- Outsourcing Platforms
- Contract Manufacturing Networks
- _Integrated Development Platforms
- Third-Party Service Providers
- Consulting & Regulatory Firms
- _Clinical Trial Supply Vendors
- Direct CDMO Contracts
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Primary Packaging
- Ophthalmic Bottles
- _Multi-Dose Dropper Systems
- _Value (USD Million) Dose Packaging
- Secondary Packaging
- Cartons & Protective Packaging
- _Cold Chain Packaging
- Primary Packaging
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Competition Analysis
- Competition Deep Dive
- Lonza Group
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Catalent Inc.
- Recipharm AB
- Siegfried Holding
- Piramal Pharma Solutions
- Thermo Fisher Scientific
- Evonik Industries
- Patheon
- Almac Group
- Corden Pharma
- Lonza Group
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Packaging
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Distribution Channel
- Figure 47: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Packaging
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 66: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Packaging
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 78: Western Europe Market Attractiveness Analysis by End Use
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 85: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Packaging
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 116: East Asia Market Attractiveness Analysis by End Use
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 122: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 123: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Packaging
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis