Pharmaceutical Fine Chemicals Market Size, Share, Growth and Forecast (2026 - 2036)
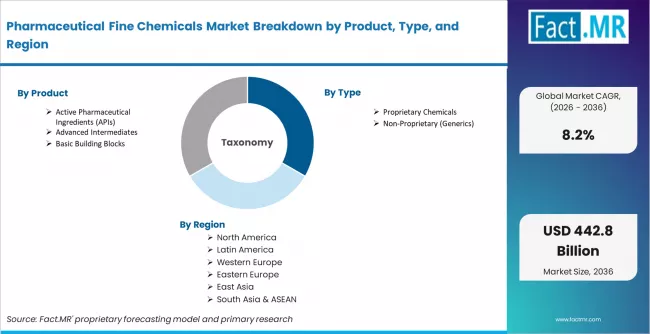
The Pharmaceutical Fine Chemicals Market is segmented by Product (Active Pharmaceutical Ingredients, Advanced Intermediates, and Basic Building Blocks), Type (Proprietary Chemicals and Non-Proprietary/Generics), Application (Oncology, Cardiovascular, and CNS and Metabolic Disorders) and Region. Forecast for 2026 to 2036.
Fact.MR indicates that the pharmaceutical fine chemicals market is at a strategic inflection point. Proprietary APIs continue to dominate the value pool. Their position is being reinforced by strong oncology pipeline growth. Competitive pressure is increasing from generics. Production of fine chemicals in India and China is accelerating. This is reshaping global supply chain economics.
Pharmaceutical Fine Chemicals Market Forecast and Outlook By Fact.MR
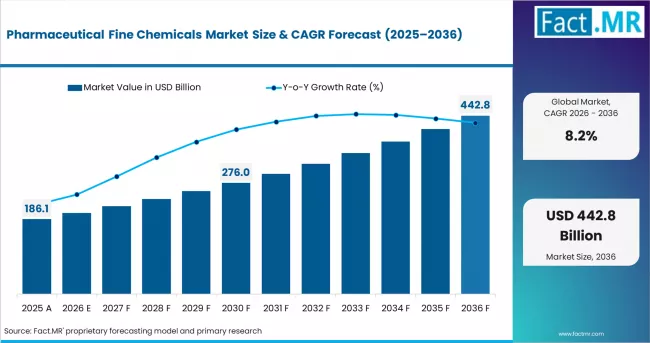
- In 2025, the pharmaceutical fine chemicals market was valued at USD 159 billion.
- Based on Fact.MR analysis, demand for pharmaceutical fine chemicals is estimated to grow to USD 172 billion in 2026 and USD 378 billion by 2036.
- FACT.MR projects a CAGR of 8.2% during the forecast period.
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 172 billion |
| Forecast Value in 2036 | USD 378 billion |
| Forecast CAGR (2026 to 2036) | 8.2% |
Summary of Pharmaceutical Fine Chemicals Market
- Market Definition
- Market covers high-purity APIs, advanced intermediates, and basic building blocks used in oncology, cardiovascular, and CNS drug manufacturing by innovator companies, generics producers, and CDMOs globally.
- Demand Drivers
- U.S. FDA drug supply security enforcement and the BIOSECURE Act are driving onshoring and dual-sourcing of API supply, increasing demand for GMP-certified fine chemical manufacturers in domestic and allied markets.
- Rapid oncology pipeline expansion, with over 1,400 oncology drugs in global clinical trials as of 2025, is increasing demand for high-potency API intermediates and specialty fine chemical synthesis capabilities.
- India's Production Linked Incentive (PLI) scheme for bulk drug parks, disbursing INR 6,940 crore through FY2024-25, is scaling certified API manufacturing capacity targeting global generic and innovator supply.
- Key Segments Analyzed
- By Product: Active Pharmaceutical Ingredients hold approximately 55% share in 2026.
- By Type: Proprietary chemicals lead with approximately 60% share in 2026 due to innovation-driven high-value API contracts.
- By Application: Oncology leads with approximately 25% share in 2026, anchored by sustained pipeline investment.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR, opines, CXOs will observe that supply chain localisation and oncology API specialization are becoming the defining competitive variables through 2036. These factors are shaping sourcing strategies and long-term partnerships.
- Strategic Implications
- Invest in GMP facility upgrades and dual-source API capability to meet FDA and EMA inspection benchmarks and secure long-term innovator and generic supply contracts.
- Target oncology API segment through proprietary synthesis development and high-potency API manufacturing infrastructure to capture premium-priced innovator pipeline contracts.
- Establish direct supply relationships with Indian and Chinese PLI-backed manufacturers to build cost-competitive intermediate sourcing and reduce raw material exposure risk.
- Methodology
- Market sizing based on API manufacturer revenues and therapeutic application volumes, validated with 2024-2025 FDA inspection records, EMA approval data, and PLI scheme disbursement reports.
- Uses U.S. FDA drug shortage database, EMA regulatory filings, India PLI scheme data, and company disclosures from BASF, Lonza, Evonik, Pfizer, and GSK.
- Forecasts incorporate oncology pipeline approval rates, generic drug demand growth, API trade flow trends, and expert inputs from CDMO and pharmaceutical supply chain specialists.
The market is set to generate an absolute opportunity of USD 219 billion between 2026 and 2036. Growth is structurally robust, anchored by persistent oncology drug demand, expanding cardiovascular generics, and CNS pipeline approvals. Expansion is partially tempered by feedstock price volatility, stringent GMP compliance costs, and geopolitical supply-chain concentration risk in key API source regions.
Procurement structures in pharmaceutical fine chemicals are becoming contract-driven and compliance-weighted. Innovator companies and CDMOs are prioritising long-term supply agreements with API manufacturers holding dual-source capabilities and validated GMP facilities. Export-oriented manufacturers in India and China are aligning facilities with U.S. FDA and EMA inspection benchmarks to secure innovator and generic contracts through 2036.
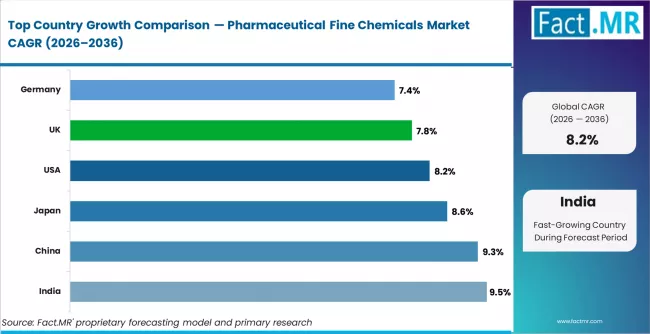
From a regional perspective, India is projected to lead with a CAGR of 9.5%, supported by PLI scheme-backed API manufacturing expansion and strong generic export demand. China follows at 9.3%, driven by domestic drug formulation volumes and specialty chemical integration. Japan registers 8.6%, reflecting high-value proprietary API demand. The United States grows at 8.2%, driven by FSMA and FDA enforcement on drug supply security. The United Kingdom, at 7.8%, is supported by post-Brexit pharmaceutical regulatory clarity and biotech supply chain development. Germany registers 7.4%, reflecting steady innovator API demand from established pharmaceutical clusters.
Segmental Analysis
Pharmaceutical Fine Chemicals Market Analysis by Product
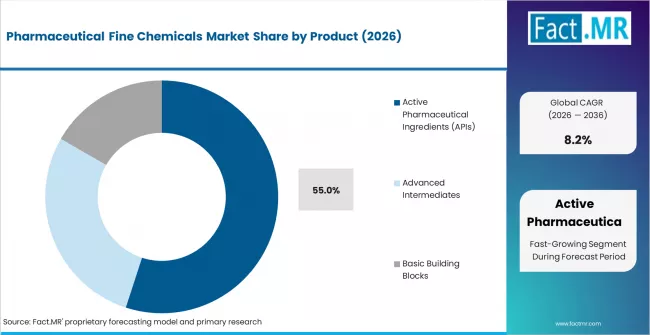
Based on FACT.MR's report, Active Pharmaceutical Ingredients are estimated to hold 55% share in 2026. APIs represent the primary regulated output of fine chemical synthesis across all therapeutic categories. Demand is driven by the expanding oncology and CNS drug pipelines, generic drug production volumes, and mandatory regulatory approval requirements tied to each API synthesis route.
- Lonza Capacity Expansion: Lonza Group AG announced expansion of its Visp, Switzerland API manufacturing campus in Q1 2025, adding 20,000 sq m of synthesis and HPAPI containment capacity to support oncology drug manufacturer supply agreements through 2030. [4]
- Evonik HPAPI Development: Evonik Industries AG launched a dedicated HPAPI synthesis service line at its Hanau, Germany facility in Q3 2024, targeting antibody-drug conjugate API demand from oncology innovator pipelines in the U.S. and Europe. [5]
- Generic API Trade Trend: India's API exports reached USD 7.8 billion in FY2024-25, per Pharmaceuticals Export Promotion Council (Pharmexcil) data, with cardiovascular, anti-infective, and CNS APIs accounting for the largest volume categories. [6]
Pharmaceutical Fine Chemicals Market Analysis by Type
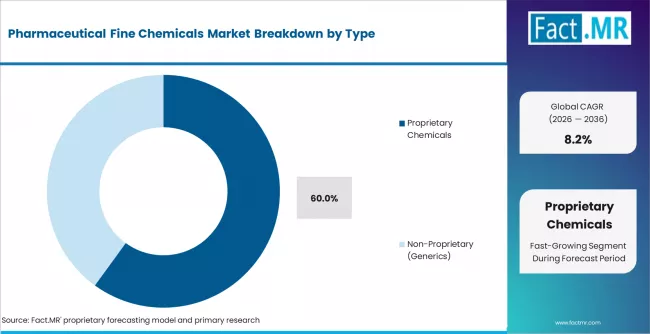
Based on FACT.MR's report, proprietary chemicals are estimated to hold 60% share in 2026. Proprietary fine chemicals generate higher per-unit revenues due to patent-protected synthesis routes and exclusive supply agreements with innovator pharmaceutical companies. The non-proprietary generics segment is the fastest-growing, driven by global off-patent drug demand and expanding CDMO generic supply contracts.
- Pfizer Proprietary API Contract: Pfizer Inc. extended its proprietary API supply agreement with a third-party fine chemical manufacturer in India in Q4 2024, covering synthesis of oncology and cardiovascular active ingredients under a multi-year GMP-validated contract framework. [7]
- GSK Generic Sourcing: GSK plc expanded its non-proprietary API sourcing from PLI-backed Indian manufacturers in H1 2025, targeting cost reduction in cardiovascular and respiratory generic drug production while maintaining dual-source qualification requirements. [8]
- Generic Growth Signal: Global generic drug approvals by the U.S. FDA reached 1,235 in FY2024, per FDA CDER annual report, driving direct demand for non-proprietary fine chemical intermediates and APIs from cost-competitive Asian manufacturers. [9]
Pharmaceutical Fine Chemicals Market Analysis by Application
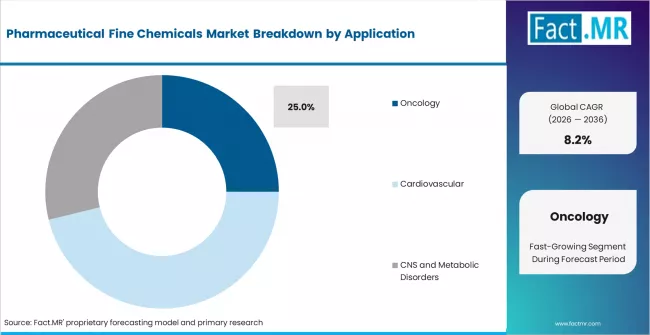
Based on FACT.MR's report, oncology is estimated to hold 25% share in 2026 across the application segment. Oncology leads due to high-volume pipeline activity, premium API pricing, and synthesis complexity that favours specialised fine chemical manufacturers. Cardiovascular and CNS segments follow, driven by large generic volumes and growing CNS pipeline approvals.
- BASF Oncology API Investment: BASF SE invested in expansion of its oncology-focused fine chemical synthesis operations at its Ludwigshafen campus in Q2 2025, adding capacity for cytotoxic API intermediates targeting immuno-oncology drug manufacturers in Europe and the United States. [10]
- CNS Pipeline Development: Albemarle Corporation expanded lithium-based CNS API intermediate supply agreements in Q4 2024, aligned with growing demand from CNS drug formulators in the U.S. and Europe following regulatory approvals for Alzheimer's and mood disorder treatments.
- Cardiovascular API Trend: The WHO Essential Medicines cardiovascular API demand estimation for 2024-2030 projects a 12% annual volume increase for generic cardiovascular fine chemicals, driven by aging demographics and expanding access in South Asia and Sub-Saharan Africa.
Drivers, Restraints, and Opportunities
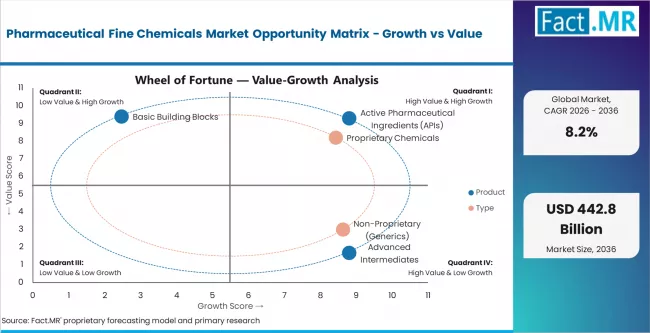
Fact.MR analysis indicates the pharmaceutical fine chemicals market operates within a structurally expanding demand environment. Growth has been sustained by continuous pharmaceutical R&D investment, global generic drug proliferation, and escalating regulatory requirements for API purity and traceability.
The market is experiencing a dual-track transition. Proprietary API demand remains high-value and innovation-driven, while the generic fine chemical segment is intensifying in volume and price competition. Standard intermediate products face margin compression.
- BIOSECURE Act Supply Shift: The U.S. BIOSECURE Act, advanced through congressional review in 2024, is accelerating decoupling of U.S. pharmaceutical supply chains from Chinese contract manufacturers. Companies such as Lonza and Evonik are expanding U.S. and European API capacity in response. This is redirecting high-value synthesis contracts toward GMP-compliant manufacturers in India, Europe, and North America. [1]
- EMA Annex 1 GMP Update: The European Medicines Agency's updated Annex 1 GMP guidelines, fully effective from August 2023 and enforced through 2024-2025 compliance cycles, are tightening contamination control requirements for sterile API production. This is increasing investment in isolator technology and advanced cleanroom systems across European and Indian API facilities aligned with EU supply contracts. [2]
- India PLI Scheme API Expansion: India's PLI Scheme for bulk drugs allocated INR 6,940 crore through FY2024-25, supporting construction of three dedicated bulk drug parks. Facilities in Himachal Pradesh, Andhra Pradesh, and Gujarat are becoming operational, targeting supply of 41 identified KSMs and APIs to domestic and global markets, reducing import dependency on China. [3]
Regional Analysis
The pharmaceutical fine chemicals market is assessed across Asia Pacific, North America, Europe, Latin America, and Middle East and Africa, covering 40+ countries with distinct demand profiles shaped by API manufacturing capability, drug pipeline activity, regulatory frameworks, and generic drug export volumes. The full report offers market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 8.2% |
| Germany | 7.4% |
| United Kingdom | 7.8% |
| China | 9.3% |
| India | 9.5% |
| Japan | 8.6% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Pharmaceutical Fine Chemicals Market Analysis
Asia Pacific is the highest-growth region, anchored by India and China as the dominant API manufacturing hubs. The region combines the world's largest generic drug production base with rapidly expanding proprietary API synthesis investment.
- China: Pharmaceutical fine chemical demand in China is projected to rise at 9.3% CAGR through 2036, driven by domestic generic drug formulation volumes and specialty API export expansion. NMPA updated drug manufacturing GMP compliance timelines in 2024, requiring certified API facilities to complete environmental and process upgrades. Zhejiang Huahai Pharmaceutical expanded its Linhai API synthesis campus in Q3 2024, adding carbapenem and cardiovascular intermediate capacity targeting Japanese and European supply agreements.
- India: Pharmaceutical fine chemical demand in India is projected to rise at 9.5% CAGR through 2036, the fastest among covered markets. India's PLI Scheme for bulk drugs, with INR 6,940 crore disbursed through FY2024-25, is building dedicated API parks in Andhra Pradesh, Himachal Pradesh, and Gujarat. DPIIT approved 41 identified KSM and API production facilities under the scheme for domestic and global supply. Pharmexcil data confirms API exports reached USD 7.8 billion in FY2024-25, with the U.S. as the largest destination.
- Japan: Japan is projected to rise at 8.6% CAGR through 2036, driven by high-value proprietary API procurement and domestic biologic drug pipeline investment. PMDA accelerated conditional approval pathways for oncology and CNS drugs in 2024, increasing demand for specialised fine chemical intermediates. Takeda Pharmaceutical expanded API supply agreements for GI oncology products with European CDMO partners in Q1 2025, representing increased cross-border fine chemical procurement.
FACT.MR's analysis of pharmaceutical fine chemicals market in Asia Pacific covers India, China, Japan, South Korea, ASEAN, and Australia and New Zealand. Readers can find API manufacturing capacity expansion trends, PLI scheme impact data, NMPA and PMDA regulatory timelines, and therapeutic application-level demand benchmarks.
North America Pharmaceutical Fine Chemicals Market Analysis
North America is the most innovation-intensive market, anchored by the U.S. as the world's largest pharmaceutical end-market and primary driver of proprietary API contract demand. The BIOSECURE Act and FDA drug supply security initiatives are reshaping sourcing strategies. Lonza and Evonik hold strong positions through GMP-certified North American API synthesis facilities and innovator supply relationships.
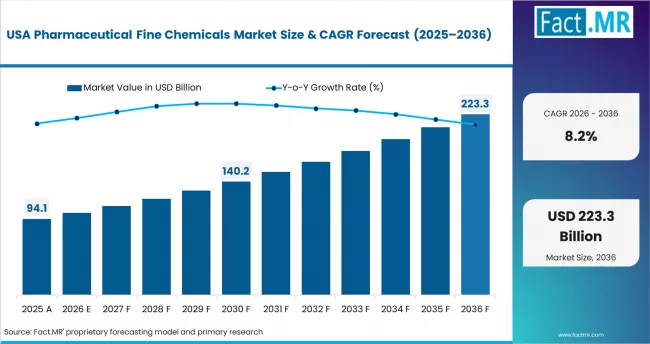
- United States: Pharmaceutical fine chemical demand in the United States is projected to rise at 8.2% CAGR through 2036. FDA's drug shortage prevention programme expanded enforcement in 2024, requiring manufacturers of critical APIs to maintain domestic or allied-nation backup supply. The BIOSECURE Act, passed through congressional markup in 2024, restricts U.S. federal drug procurement from named Chinese CDMOs, accelerating sourcing shifts to India and domestic producers.
FACT.MR's analysis of pharmaceutical fine chemicals market in North America covers the United States, Canada, and Mexico. Readers can find BIOSECURE Act supply chain impact data, FDA approval benchmarks, oncology API procurement trends, and CDMO contract dynamics.
Europe Pharmaceutical Fine Chemicals Market Analysis
Europe is a regulation-intensive, innovation-anchored market defined by EMA's Annex 1 GMP enforcement, the EU Pharmaceutical Strategy, and leading innovator pharmaceutical clusters in Germany, Switzerland, the UK, and Ireland. BASF, Evonik, and Lonza hold structural advantages through integrated synthesis, EU-certified GMP facilities, and direct innovator supply relationships.
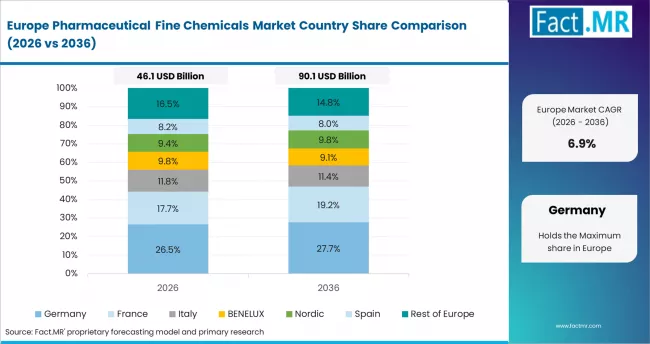
- Germany: Pharmaceutical fine chemical demand in Germany is projected to rise at 7.4% CAGR through 2036. EMA's revised Annex 1 GMP guidelines, enforced through 2024-2025 compliance cycles, are increasing capital investment in sterile API manufacturing infrastructure across German facilities. BASF expanded its specialty fine chemical synthesis operations at Ludwigshafen in Q2 2025, targeting oncology and CNS API intermediates.
- United Kingdom: Pharmaceutical fine chemical demand in the United Kingdom is projected to rise at 7.8% CAGR through 2036. The MHRA aligned its GMP inspection framework with the PICS scheme in Q1 2025, enabling more efficient bilateral API manufacturer qualification between the UK and international markets post-Brexit. GSK plc expanded API sourcing agreements with UK-qualified Indian and European fine chemical manufacturers in H1 2025 to reduce post-Brexit supply dependency risk.
FACT.MR's analysis of pharmaceutical fine chemicals market in Europe covers Germany, France, the UK, Italy, Spain, and Rest of Europe. Readers can find EMA Annex 1 compliance impact data, EU Pharma Strategy supply chain investment trends, and proprietary API procurement benchmarks by therapeutic category.
Competitive Aligners for Market Players
Competition in the pharmaceutical fine chemicals market is shifting toward capability depth and supply chain control. Vendors must move beyond basic chemical synthesis and position themselves as strategic partners to innovator pharma.
Oncology API specialisation is the first critical lever. High-growth oncology pipelines are driving demand for complex, high-purity intermediates. Companies with advanced synthesis capabilities and regulatory approvals are securing long-term contracts. This segment commands premium margins and stable demand. Supply chain localisation is becoming essential. Regulatory frameworks in the U.S. and Europe are pushing for domestic or allied sourcing. Dual-source capability is now a baseline requirement. Companies must offer geographically diversified production to mitigate supply risk.
CDMO integration is a key differentiator. Players such as Lonza Group and Evonik Industries are strengthening positions through end-to-end manufacturing services. Integrated offerings improve client retention and increase contract value.
Cost competitiveness remains important in generics. Manufacturers in India and China are scaling production efficiently. This is reshaping pricing dynamics across non-proprietary segments. Zhejiang Huahai Pharmaceutical exemplifies scale-driven expansion strategies. Regulatory compliance and GMP certification are non-negotiable. Increasing inspection intensity is raising entry barriers. Vendors must invest in validated facilities and documentation systems to remain competitive. Technology adoption is also emerging as a differentiator. Continuous manufacturing and process optimisation reduce cost and improve consistency. These capabilities are gaining importance in both proprietary and generic segments.
Key Players
- BASF SE
- Lonza Group AG
- Evonik Industries AG
- Albemarle Corporation
- Pfizer Inc.
- GSK plc
- Boehringer Ingelheim GmbH
- Siegfried Holding AG
- Cambrex Corporation
- Dishman Carbogen Amcis
Bibliography
- [1] U.S. Congress. (2024). BIOSECURE Act: Proposed Restrictions on Federal Procurement from Chinese Biotechnology Companies Including WuXi AppTec and BGI. congress.gov
- [2] European Medicines Agency. (2023, August; enforced 2024-2025). Annex 1: Manufacture of Sterile Medicinal Products. ema.europa.eu
- [3] Department of Pharmaceuticals, Government of India. (FY2024-25). PLI Scheme for Bulk Drugs: INR 6,940 Crore Disbursement and Bulk Drug Park Progress Report. pharmaceuticals.gov.in
- [4] Lonza Group AG. (Q1 2025). Press Release: Expansion of Visp API and HPAPI Manufacturing Campus for Oncology and ADC Intermediate Supply. lonza.com/media
- [5] Evonik Industries AG. (Q3 2024). Press Release: Launch of HPAPI Synthesis Service Line at Hanau for Antibody-Drug Conjugate API Demand. evonik.com/news
- [6] Pharmaceuticals Export Promotion Council of India (Pharmexcil). (FY2024-25). Annual API Export Data Report: USD 7.8 Billion API Exports with Category-Wise Breakdown. pharmexcil.com
- [7] Pfizer Inc. (Q4 2024). Investor Disclosure: Multi-Year GMP-Validated Proprietary API Supply Agreement with India-Based Fine Chemical Manufacturer for Oncology and Cardiovascular Products. pfizer.com/investors
- [8] GSK plc. (H1 2025). Annual Report Supplement: Non-Proprietary API Sourcing Expansion from PLI-Backed Indian Manufacturers for Cardiovascular and Respiratory Generic Portfolio. gsk.com/investors
- [9] U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (FY2024). CDER Annual Report: Generic Drug Approvals FY2024. fda.gov/drugs/cder
- [10] BASF SE. (Q2 2025). Press Release: Oncology Fine Chemical Synthesis Expansion at Ludwigshafen for Cytotoxic API Intermediates Targeting Immuno-Oncology Supply. basf.com/agriculture
This Report Addresses
- Strategic intelligence on pharmaceutical fine chemical demand across APIs, advanced intermediates, and basic building blocks for oncology, cardiovascular, and CNS drug manufacturing globally.
- Market forecast from USD 172 billion in 2026 to USD 378 billion by 2036, at a CAGR of 8.2%.
- Growth opportunity mapping across India PLI scheme API expansion, U.S. BIOSECURE Act supply chain reshoring, China API manufacturing capacity growth, and Germany EMA Annex 1 compliance investment.
- Segment analysis by product (APIs, advanced intermediates, basic building blocks), type (proprietary, generics), and application (oncology, cardiovascular, CNS) across global and regional markets.
- Regional outlook covering Asia Pacific API manufacturing leadership, North America innovation-driven demand, Europe EMA compliance-driven procurement, and emerging market generic expansion.
- Competitive landscape of BASF, Lonza, Evonik, Albemarle, Pfizer, and GSK covering GMP certification depth, HPAPI synthesis capability, and long-term innovator supply contract positioning.
- Regulatory impact analysis covering U.S. BIOSECURE Act supply security requirements, EMA Annex 1 GMP guidelines, India PLI scheme API park development, and NMPA GMP compliance timelines.
- Report delivered in PDF, Excel datasets, and presentation formats, supported by primary interviews, API manufacturer filings, and drug approval and trade flow data.
Pharmaceutical Fine Chemicals Market Definition
The pharmaceutical fine chemicals market encompasses high-purity chemical compounds used as active pharmaceutical ingredients (APIs), advanced intermediates, and basic building blocks in drug manufacturing.
Pharmaceutical Fine Chemicals Market Inclusions
Covers global and regional forecasts from 2026 to 2036 by product, type, and application. Includes APIs, advanced intermediates, and basic building blocks. Covers proprietary and non-proprietary chemical types. Applications span oncology, cardiovascular, and CNS and metabolic disorders. Encompasses CDMO supply, innovator contracts, and generic fine chemical trade flows.
Pharmaceutical Fine Chemicals Market Exclusions
Excludes finished dosage forms such as tablets, capsules, and injectables. Omits bulk pharmaceutical excipients not classified as active chemical compounds. Excludes veterinary fine chemicals and agrochemical intermediates unless used in dual-approved pharmaceutical synthesis pathways.
Pharmaceutical Fine Chemicals Market Methodology
- Primary Research:Interviews with API manufacturers, CDMO procurement heads, innovator pharmaceutical sourcing managers, and regulatory compliance specialists across India, the United States, Germany, China, and Japan.
- Desk Research:Analysis of regulatory filings from BASF, Lonza, Evonik, Pfizer, and GSK, alongside U.S. FDA drug shortage database records, EMA API approval updates, and India PLI scheme disbursement data (2024-2025).
- Market-Sizing and Forecasting:Hybrid model combining top-down revenue estimates from API manufacturers with bottom-up volume estimates by application and therapeutic category, cross-referenced with average selling prices by product type.
- Data Validation and Update Cycle:Validated through CDMO procurement benchmarks, API trade flow records, regulatory inspection outcomes, and expert interviews with pharmaceutical supply chain specialists. Updated annually.
Scope of the Report
| Attribute | Details |
|---|---|
| Quantitative Units | USD 172 billion (2026) to USD 378 billion (2036), at a CAGR of 8.2% |
| Market Definition | High-purity APIs, advanced intermediates, and basic building blocks used in oncology, cardiovascular, and CNS drug manufacturing by innovator companies, generic producers, and CDMOs globally. |
| Product Segmentation | Active Pharmaceutical Ingredients, Advanced Intermediates, Basic Building Blocks |
| Type Segmentation | Proprietary Chemicals, Non-Proprietary (Generics) |
| Application Segmentation | Oncology, Cardiovascular, CNS and Metabolic Disorders |
| Application Coverage | Oncology drug API synthesis, cardiovascular generic fine chemical supply, CNS and metabolic disorder intermediate production, CDMO proprietary and generic contract manufacturing. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, ASEAN, Australia and New Zealand, Brazil, Argentina, Saudi Arabia, UAE, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | BASF SE, Lonza Group AG, Evonik Industries AG, Albemarle Corporation, Pfizer Inc., GSK plc, Boehringer Ingelheim GmbH, Siegfried Holding AG, Cambrex Corporation, Dishman Carbogen Amcis |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using API manufacturer revenues, therapeutic application volumes, average selling prices by product type, CDMO contract benchmarks, and primary interviews with pharmaceutical supply chain specialists. |
Pharmaceutical Fine Chemicals Market by Segments
-
By Product:
- Active Pharmaceutical Ingredients (APIs)
- Advanced Intermediates
- Basic Building Blocks
-
By Type:
- Proprietary Chemicals
- Non-Proprietary (Generics)
-
By Application:
- Oncology
- Cardiovascular
- CNS and Metabolic Disorders
-
By Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- UAE
- Turkey
- South Africa
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the global pharmaceutical fine chemicals market in 2025?
The global pharmaceutical fine chemicals market was valued at USD 159 billion in 2025.
What will the market size be in 2026?
Based on Fact.MR analysis, demand for pharmaceutical fine chemicals is estimated to grow to USD 172 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 378 billion by 2036, generating USD 219 billion in absolute dollar opportunity over the forecast period.
What is the expected CAGR from 2026 to 2036?
FACT.MR projects a CAGR of 8.2% for the global pharmaceutical fine chemicals market during the 2026 to 2036 forecast period.
Which product segment is poised to lead the market?
Active Pharmaceutical Ingredients (APIs) lead with approximately 55% share in 2026, reflecting their primacy as the regulated active output across all therapeutic categories.
Which type segment shows the fastest growth?
Non-proprietary generic fine chemicals represent the fastest-growing type segment, driven by global off-patent drug demand and expanding CDMO generic supply contracts in India, China, and Europe.
Which application segment holds the largest share?
Oncology leads with approximately 25% share in 2026, driven by sustained pipeline investment, high per-unit API values, and stringent synthesis purity requirements.
Which country shows the fastest growth?
India leads at 9.5% CAGR through 2036, supported by PLI scheme-backed API park development, Pharmexcil export growth to USD 7.8 billion in FY2024-25, and large-scale FDA-approved generic API manufacturing capacity.
Who are the key players in the market?
Major players include BASF SE, Lonza Group AG, Evonik Industries AG, Albemarle Corporation, Pfizer Inc., GSK plc, Boehringer Ingelheim GmbH, Siegfried Holding AG, Cambrex Corporation, and Dishman Carbogen Amcis.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Active Pharmaceutical Ingredients (APIs)
- Advanced Intermediates
- Basic Building Blocks
- Active Pharmaceutical Ingredients (APIs)
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2026 to 2036
- Proprietary Chemicals
- Non-Proprietary (Generics)
- Proprietary Chemicals
- Y to o to Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Oncology
- Cardiovascular
- CNS and Metabolic Disorders
- Oncology
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Type
- By Application
- Competition Analysis
- Competition Deep Dive
- BASF SE
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Lonza Group AG
- Evonik Industries AG
- Albemarle Corporation
- Pfizer Inc.
- GSK plc
- Boehringer Ingelheim GmbH
- Siegfried Holding AG
- Cambrex Corporation
- Dishman Carbogen Amcis
- BASF SE
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Type
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Type
- Figure 29: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Application
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product
- Figure 36: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Type
- Figure 39: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Application
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product
- Figure 46: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Type
- Figure 49: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by Application
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Type
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Application
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product
- Figure 66: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Type
- Figure 69: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by Application
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis