Medical Polyoxymethylene Market Size, Share, Growth and Forecast (2026 - 2036)
The Medical Polyoxymethylene Market is segmented by Type (Copolymer POM and Homopolymer POM), Application (Insulin Pens, Dialysis Machines, Surgical Instruments, and Inhalers) and Region. Forecast for 2026 to 2036.
FACT.MR opines that the medical POM market is approaching a clear specification inflection point, where conventional engineering-grade polyoxymethylene is steadily being replaced by high-purity, medical-grade formulations. Demand is increasingly shifting toward USP Class VI and ISO 10993-certified copolymer variants, particularly among drug delivery OEMs and surgical device manufacturers. This transition is being driven by tightening global expectations around biocompatibility, extractables and leachables control, and full material traceability.
Medical Polyoxymethylene Market Forecast and Outlook By Fact.MR
In 2025, the medical polyoxymethylene market was valued at USD 1.5 billion. Based on Fact.MR analysis, demand for medical POM is estimated to grow to USD 1.6 billion in 2026 and USD 3.7 billion by 2036. FACT.MR projects a CAGR of 8.6% during the forecast period.
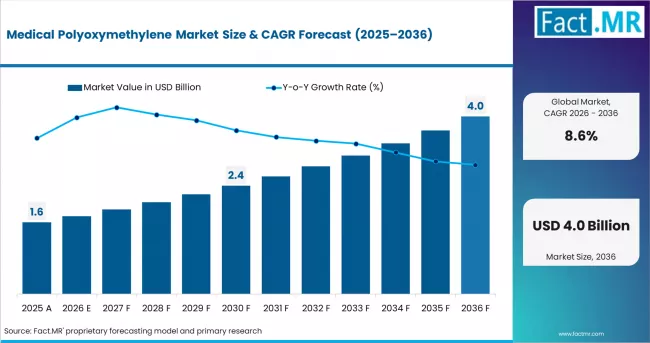
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 1.6 billion |
| Forecast Value in 2036 | USD 3.7 billion |
| Forecast CAGR (2026 to 2036) | 8.6% |
Summary of the Medical Polyoxymethylene Market
- Market Definition:
- Covers medical-grade POM copolymer and homopolymer resins with biocompatibility certification, supplied for insulin pens, dialysis systems, surgical instruments, and inhaler components to device manufacturers globally.
- Demand Drivers:
- India’s Ayushman Bharat and PLI schemes are boosting domestic device manufacturing. This is increasing demand for certified POM in insulin pens and components.
- China’s expansion in insulin pen and diabetes device production is sustaining high-volume procurement of medical-grade POM through OEM supply chains.
- EU MDR requirements are pushing manufacturers to adopt certified copolymer POM. Full ISO 10993 documentation is becoming mandatory.
- Key Segments Analyzed:
- By Type: Copolymer POM leads 63% in 2026 due to superior hydrolysis resistance and broad biocompatibility certification acceptance by drug delivery OEMs.
- By Application: Insulin Pens lead 30% in 2026, driven by global diabetes device production growth and high POM component content per device unit.
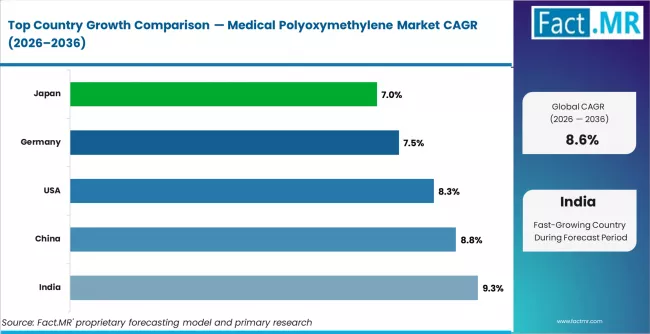
- By Geography: India grows fastest at 9.3% CAGR driven by domestic medical device manufacturing investment and healthcare infrastructure expansion.
- Analyst Opinion at FACT.MR:
- Shambhu Nath Jha, Principal Consultant at Fact.MR, states that biocompatibility certification is the primary qualification barrier. Medical POM must meet USP Class VI and ISO 10993 standards. Full traceability documentation is essential. Suppliers with certified compliance gain a clear advantage. Drug delivery OEMs prefer validated material sources. Long-term supply agreements are common. These suppliers capture structural margin premiums.
- Strategic Implications:
- Invest in USP Class VI and ISO 10993-certified copolymer POM to qualify for drug delivery OEM programmes in India, China, and the United States.
- Develop low-formaldehyde migration POM grades to meet EU MDR biocompatibility requirements in surgical and inhaler applications.
- Scale certified medical-grade POM capacity to reduce costs and compete in insulin pen and dialysis procurement.
- Methodology:
- Market sizing is based on device production volumes and POM usage assumptions by application. Validated through producer revenues and OEM procurement benchmarks.
- Supported by EU MDR compliance data, India PLI scheme records, FDA 510(k) data, and China device production statistics.
- Forecasts incorporate device pipelines, qualification timelines, pricing benchmarks, and expert interviews with medical polymer specialists.
The market is expected to generate approximately USD 2.1 billion in incremental revenue over the forecast period. This reflects transformational growth driven by rising insulin pen and inhaler device production in Asia Pacific, surgical instrument localisation in emerging markets, and expanding dialysis infrastructure globally. Growth is constrained by biocompatibility certification timelines, formaldehyde migration compliance under EU MDR, and limited medical-grade POM producer qualification base.
Medical POM procurement is shifting from standard polymer supply contracts toward specification-locked supply agreements tied to device-specific biocompatibility validation. Drug delivery OEMs require documented USP Class VI and ISO 10993 test data, and any formulation change triggers full re-qualification. This creates durable supplier relationships and supports premium pricing for certified medical-grade copolymer POM relative to industrial grades.
India leads country-level growth at 9.3% CAGR through 2036, driven by domestic medical device manufacturing expansion and healthcare infrastructure investment. China follows at 8.8%, supported by the world's largest insulin pen and medical device production base. The United States records 8.3%, anchored by advanced drug delivery system demand and precision surgical instrument procurement. Germany records 7.5%, driven by medical engineering innovation and EU MDR biocompatibility compliance upgrades. Japan records 7.0%, focused on high-performance medical-grade polymer supply for precision device manufacturing, with mature markets constrained by long re-qualification cycles and limited new-device volume upside.
Segmental Analysis
Medical Polyoxymethylene Market Analysis by Type
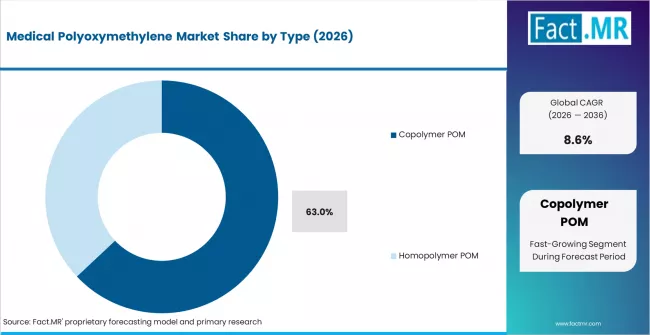
Copolymer POM holds 63% share in 2026. It offers better thermal stability and hydrolysis resistance. Certification coverage is broader than homopolymer grades. OEMs prefer it for consistent performance in sterilization cycles.
- Celanese Medical Grade Expansion: Celanese Corporation expanded Hostaform medical-grade copolymer POM in 2024. New grades target insulin pen and inhaler components. This supports ISO 10993 qualification needs. [4]
- BASF Ultraform Development: BASF SE launched an updated Ultraform grade in 2025. It targets low-formaldehyde migration and EU MDR compliance. [2]
- Copolymer Certification Trend: Medical polymer certification bodies reported a 22% rise in POM test submissions in 2024. Growth is driven by EU MDR upgrades and Asia Pacific demand. [5]
Medical Polyoxymethylene Market Analysis by Application
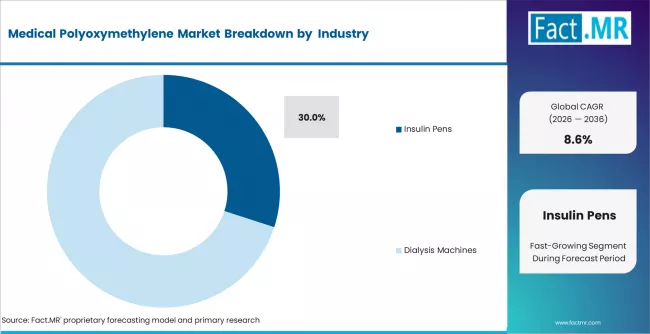
Insulin pens account for 30% share in 2026. Demand is driven by global diabetes device expansion. POM is used in high-precision components requiring stability and resistance.
- Polyplastics Insulin Pen Programme: Polyplastics Co., Ltd. expanded supply to insulin pen OEMs in 2024. It offers USP Class VI certified grades with technical support. [6]
- Mitsubishi Dialysis Grade Development: Mitsubishi Engineering-Plastics Corporation launched a dialysis-grade POM in 2025. It supports chemical resistance and steam sterilisation. [7]
- Inhaler Component Adoption Trend: The European Pharmaceutical Aerosol Group reported 18% growth in 2024. Medical-grade POM is used in inhaler mechanisms requiring precision and stability. [8]
Drivers, Restraints, and Opportunities
FACT.MR analysts observe that the medical POM market has moved from a niche segment to an early-growth phase. Growth is driven by expansion in diabetes devices and surgical instruments. Demand is anchored in insulin pen scale-up in Asia Pacific and dialysis infrastructure globally.
The market is shifting toward certification-led supply. Industrial-grade POM is losing share in regulated applications. Certified medical-grade copolymer POM is gaining adoption. Price premiums of 25–40% are common over standard grades.
- India Medical Device PLI Scheme: India’s PLI scheme is driving local device manufacturing investment. Polyplastics and Celanese expanded medical-grade POM supply to Indian OEMs. This creates a new high-volume demand base. [1]
- EU MDR Biocompatibility Driver: EU Medical Device Regulation (EU MDR 2017/745) mandates ISO 10993 testing for patient-contact materials. Surgical and inhaler manufacturers are shifting to certified copolymer POM. BASF expanded its Ultraform certification programme. [2]
- China Diabetes Device Scale-up: China approved multiple insulin pen platforms, increasing domestic production. Polyplastics and Mitsubishi Engineering-Plastics Corporation reported higher regional supply volumes. This is the largest volume growth driver globally. [3]
Regional Analysis
The medical POM market is assessed across Asia Pacific, North America, Europe, Latin America, and Middle East and Africa, covering 40+ countries with distinct demand profiles shaped by medical device production scale, healthcare infrastructure investment, biocompatibility regulation stringency, and drug delivery device procurement frameworks. The full report provides market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.3% |
| China | 8.8% |
| United States | 8.3% |
| Germany | 7.5% |
| Japan | 7.0% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Medical POM Market Analysis
Asia Pacific is the primary production base for medical POM, supported by strong device manufacturing ecosystems and policy-led localisation across key economies. China and India anchor large-scale insulin pen and drug delivery production, while Japan and South Korea contribute through high-precision medical polymer applications.
- China: China remains the dominant volume centre with consumption projected at 8.8% CAGR through 2036. Growth is closely linked to the scale of domestic diabetes device and surgical instrument manufacturing under National Medical Products Administration frameworks. Approvals for multiple insulin pen platforms have increased component output, directly raising POM usage in precision parts. Procurement is highly structured, with OEMs relying on pre-qualified suppliers capable of delivering certified materials with consistent quality and long-term supply assurance rather than competing on price.
- India: India reflects policy-driven acceleration at 9.3% CAGR through 2036. Government-backed PLI schemes are enabling domestic manufacturing of insulin pens, inhalers, and surgical components. Regulatory alignment through BIS updates is introducing ISO 10993-linked documentation requirements, shifting procurement away from unbranded materials toward certified copolymer POM. This transition is formalising a new demand base tied to public procurement and export-oriented device production.
FACT.MR analysis covers China, Japan, South Korea, India, ASEAN, and Australia and New Zealand. It includes device production trends, certification benchmarks, and country-level forecasts.
Europe Medical POM Market Analysis
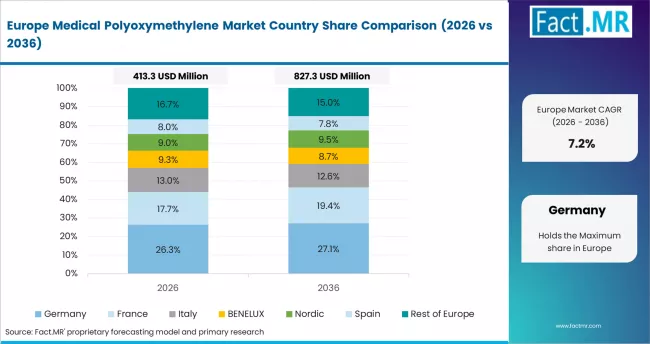
Europe acts as a regulatory benchmark for biocompatibility compliance and precision medical engineering. EU MDR frameworks define material qualification standards across surgical instruments, dialysis systems, and inhaler components. BASF, Celanese, and SABIC support certified intermediate supply through extensive compliance documentation and application-specific grades.
- Germany: Germany shows regulation-aligned progression at 7.5% CAGR through 2036. Its advanced medical engineering base drives structured procurement of high-performance polymers. EU MDR enforcement, along with updated national guidance on formaldehyde migration limits, is tightening acceptance criteria for patient-contact materials.
FACT.MR analysis covers Germany, France, the UK, Italy, Spain, and Rest of Europe. It includes regulatory trends, compliance benchmarks, and forecasts.
North America Medical POM Market Analysis
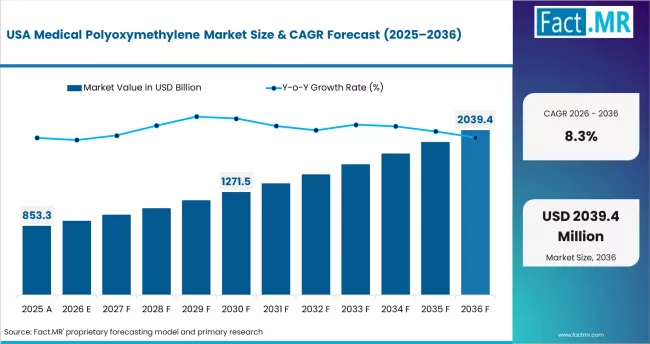
North America represents a compliance- and innovation-led environment anchored in advanced drug delivery systems and surgical devices. Regulatory frameworks, combined with strong OEM engineering capabilities, shape material selection and qualification cycles. Celanese Corporation and DuPont maintain strong supply positions through certified medical-grade POM portfolios and technical collaboration with device manufacturers. The region also shows increasing focus on supply chain resilience and domestic sourcing.
- United States: The United States indicates qualification-driven expansion at 8.3% CAGR through 2036. FDA 510(k) pathways require full ISO 10993 documentation for polymer materials used in medical devices. This creates recurring material qualification cycles as device designs evolve. At the same time, innovation in insulin pens, autoinjectors, and minimally invasive devices is increasing reliance on high-performance, certified POM grades, while policy support for domestic manufacturing is strengthening local supply chains.
FACT.MR analysis covers the United States, Canada, and Mexico. It includes FDA compliance data, device production trends, and country-level forecasts.
Competitive Aligners for Market Players
The medical POM market reflects a dual-layer competitive structure, where a limited group of global engineering polymer producers dominate certified medical-grade supply, while a wider base of industrial POM manufacturers remains largely confined to non-regulated applications. Companies such as Celanese Corporation, BASF SE, Polyplastics Co., Ltd., and Mitsubishi Engineering-Plastics Corporation hold leading positions due to their extensive portfolios of certified medical-grade POM. These suppliers have built capabilities around USP Class VI, ISO 10993, and EU MDR compliance, which are essential for participation in regulated medical device applications.
Competitive advantage in this space is closely tied to certification depth and established qualification status with OEMs. For instance, Celanese’s Hostaform and BASF’s Ultraform medical-grade POM ranges are approved across multiple regulatory systems, including those governed by the FDA and European authorities. This multi-region compliance allows them to serve global device manufacturers without requiring reformulation or revalidation. Replicating such certification coverage involves high cost, long timelines, and technical complexity, which limits new entrants and reduces pricing pressure in this segment.
Procurement dynamics further reinforce this structure. Medical device manufacturers typically qualify specific POM grades for each product platform through detailed biocompatibility validation processes. Once approved, switching suppliers is not straightforward, as it requires complete requalification under regulatory frameworks, often taking 12 to 24 months. As a result, supplier relationships tend to be long-term and stable. This creates a market environment where certified suppliers benefit from consistent demand, stronger margins, and limited competition from lower-cost, non-certified alternatives.
Key Players
- Celanese Corporation
- Polyplastics Co., Ltd.
- BASF SE
- DuPont
- Mitsubishi Engineering-Plastics Corporation
- SABIC
- Asahi Kasei Corporation
Bibliography
- [1] Ministry of Chemicals and Fertilizers, Government of India. (2024). Production Linked Incentive Scheme for Medical Devices: Investment Milestone Achievement and Domestic Medical Device Manufacturing Expansion Including Drug Delivery Component Production Data. 2024. pharmaceuticals.gov.in
- [2] BASF SE. (2024). Ultraform Medical POM Certification Programme Expansion: ISO 10993 and EU MDR Compliance Documentation Update for Surgical Instrument and Inhaler Component Applications in European Device Manufacturing Markets. 2024. basf.com/investor-relations
- [3] National Medical Products Administration, China. (2024). Medical Device Registration Approvals: 14 New Insulin Pen Platform Approvals Including Component Material Specification Requirements and Domestic Manufacturing Partner Documentation. 2024. nmpa.gov.cn
- [4] Celanese Corporation. (2024). Hostaform Medical Grade POM Portfolio Expansion: New USP Class VI and ISO 10993-Certified Copolymer Grades for Insulin Pen Cartridge and Autoinjector Mechanism Applications. 2024. celanese.com/investor-relations
- [5] International Organization for Standardization. (2024). ISO 10993 Medical Device Biocompatibility Testing Submission Data 2024: Polymer Test Submission Volume Growth Including POM Formaldehyde Migration and Cytotoxicity Testing for EU MDR and FDA Compliance. 2024. iso.org
- [6] Polyplastics Co., Ltd. (2024). Medical Grade POM Supply Programme Expansion: USP Class VI Certified Copolymer Resin Supply Agreements with Insulin Pen and Medical Device OEMs in Japan and China. 2024. polyplastics.com/news
- [7] Mitsubishi Engineering-Plastics Corporation. (2025). New Product Launch: Dialysis-Grade POM Resin with Enhanced Chemical Resistance and Long-Term Steam Sterilisation Compatibility Certification for Dialysis Machine Fluid Pathway Component Applications. January 2025. m-ep.co.jp/news
- [8] European Pharmaceutical Aerosol Group. (2024). Precision Polymer Demand in Dry Powder Inhaler Manufacturing Report 2024: Medical-Grade POM Adoption Growth for Dose Counter and Valve Housing Components Under EU Inhaler Device Regulatory Standards. 2024. epag.eu
This Report Addresses
- Strategic insights on medical POM demand across India PLI-driven device manufacturing expansion, China NMPA insulin pen device approvals, U.S. FDA 510(k) biocompatibility certification programmes, Germany EU MDR compliance upgrades, and Japan PMDA precision polymer requirements.
- Market forecast from USD 1.6 billion in 2026 to USD 3.7 billion by 2036 at a CAGR of 8.6%.
- Growth opportunity mapping across insulin pen and drug delivery OEM copolymer POM qualification pipelines, dialysis machine infrastructure procurement expansion, surgical instrument medical-grade polymer upgrade demand, and inhaler precision component supply.
- Segment analysis by POM type (copolymer, homopolymer) and application (insulin pens, dialysis machines, surgical instruments, inhalers).
- Regional outlook covering Asia Pacific device production volume leadership, North America FDA compliance-driven drug delivery procurement, and Europe EU MDR-led biocompatibility upgrade demand cycles.
- Competitive analysis of Celanese Corporation, Polyplastics, BASF SE, DuPont, Mitsubishi Engineering-Plastics, SABIC, and Asahi Kasei covering certification portfolio, OEM qualification status, and application-specific supply positioning.
- Regulatory impact analysis covering EU MDR ISO 10993 biocompatibility mandate, India PLI medical device manufacturing incentive, U.S. FDA 510(k) polymer documentation requirements, and Japan PMDA formaldehyde migration compliance standards.
- Report available in PDF, Excel datasets, and presentation formats supported by primary medical POM producer interviews, FDA device approval data, EU MDR compliance filings, and India PLI scheme government programme records.
Medical Polyoxymethylene Market Definition
The medical POM market comprises biocompatibility-certified polyoxymethylene copolymer and homopolymer resins meeting USP Class VI and ISO 10993 standards. These materials are used in precision drug delivery systems, surgical components, dialysis fittings, and inhaler mechanisms requiring high dimensional stability, chemical resistance, and low friction.
Medical Polyoxymethylene Market Inclusions
Covers global and regional demand from 2026 to 2036 by POM type and medical application. Includes certified copolymer and homopolymer POM used in insulin pens, dialysis systems, surgical instruments, and inhalers across Asia Pacific, North America, and Europe.
Medical Polyoxymethylene Market Exclusions
Excludes non-certified engineering-grade and industrial POM. Finished medical devices are not included. Also excludes POM used in automotive, electronics, and other non-medical applications. Focus remains on medical-grade resin supply to device manufacturers.
Medical Polyoxymethylene Market Research Methodology
- Primary Research: Includes interviews with medical POM producers, drug delivery OEM procurement teams, surgical device manufacturers, and biocompatibility specialists.
- Desk Research: Covers analysis of Celanese, BASF, and Polyplastics reports, EU MDR compliance data, FDA 510(k) records, and recent polymer supply chain studies.
- Market Sizing and Forecasting: Uses a hybrid model based on device production volumes, POM usage per device, pricing by certification grade, and policy-driven demand for insulin and dialysis devices.
- Data Validation and Update Cycle: Validated through producer revenues, OEM procurement data, certification records, and expert inputs. Updated with new medical device programme developments.
Scope of Report
| Attribute | Details |
|---|---|
| Quantitative Units | USD 1.6 billion (2026) to USD 3.7 billion (2036), at a CAGR of 8.6% |
| Market Definition | Medical-grade POM copolymer and homopolymer resins certified under USP Class VI and ISO 10993, supplied for insulin pen, dialysis machine, surgical instrument, and inhaler component manufacturing globally. |
| Type Segmentation | Copolymer POM, Homopolymer POM |
| Application Segmentation | Insulin Pens, Dialysis Machines, Surgical Instruments, Inhalers |
| Application Coverage | Insulin pen cartridge and dose mechanism components, dialysis machine fluid pathway and fitting supply, surgical instrument handle and joint part manufacturing, and dry powder inhaler dose counter and valve housing applications. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, ASEAN, Australia and New Zealand, Brazil, Argentina, Chile, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | Celanese Corporation, Polyplastics Co. Ltd., BASF SE, DuPont, Mitsubishi Engineering-Plastics Corporation, SABIC, Asahi Kasei Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using medical device production volumes, POM content assumptions by device type, average selling prices by certification grade, biocompatibility qualification timelines, and primary interviews with medical POM producers, drug delivery OEM procurement teams, and device biocompatibility specialists. |
Medical Polyoxymethylene Market by Segments
-
By Type:
- Copolymer POM
- Homopolymer POM
-
By Application:
- Insulin Pens
- Dialysis Machines
- Surgical Instruments
- Inhalers
-
By Region:
-
North America
- United States
- Canada
- Mexico
-
Latin America
- Brazil
- Mexico
- Argentina
- Chile
- Rest of Latin America
-
Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
-
Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
-
East Asia
- China
- Japan
- South Korea
-
South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
-
Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East and Africa
-
- Frequently Asked Questions -
How large is the medical POM market in 2025?
The market was valued at USD 1.5 billion in 2025.
What will the market size be in 2026?
The market is estimated to reach USD 1.6 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 3.7 billion by 2036.
What is the expected CAGR of the medical POM market?
The market is expected to grow at a CAGR of 8.6% from 2026 to 2036.
What is the absolute dollar opportunity in this market?
The market is expected to generate approximately USD 2.1 billion in incremental revenue between 2026 and 2036.
Which POM type segment leads the market?
Copolymer POM leads with approximately 63% share in 2026, driven by superior biocompatibility certification coverage and hydrolysis resistance.
Which application segment holds the largest share?
Insulin Pens lead with approximately 30% share in 2026, driven by global diabetes device production growth.
Which country shows the fastest growth?
India leads with a CAGR of 9.3% through 2036, driven by domestic medical device manufacturing expansion under the PLI scheme.
What is medical-grade POM?
Medical-grade POM is a biocompatibility-certified polyoxymethylene resin meeting USP Class VI and ISO 10993 standards, used for precision drug delivery, dialysis, surgical, and inhaler components requiring dimensional stability and chemical resistance.
What is driving market growth?
Growth is driven by insulin pen and medical device production scale-up in Asia Pacific, EU MDR biocompatibility upgrade demand, U.S. FDA drug delivery device procurement, and India PLI-driven domestic manufacturing investment.
Why do certified POM suppliers hold structural pricing advantages?
Device-level biocompatibility qualification creates high switching costs, as replacing an approved POM grade triggers full ISO 10993 and regulatory revalidation, insulating certified suppliers from price competition in OEM supply programmes.
Which end-use application dominates?
Insulin pens hold the leading application position at 30% share in 2026, driven by the expanding global diabetes device production base and high POM component content per device unit.
Who are the key players?
Leading companies include Celanese Corporation, Polyplastics Co., Ltd., BASF SE, DuPont, Mitsubishi Engineering-Plastics Corporation, SABIC, and Asahi Kasei Corporation.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Copolymer POM
- Homopolymer POM
- Copolymer POM
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Insulin Pens
- Dialysis Machines
- Surgical Instruments
- Insulin Pens
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Celanese Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Polyplastics Co., Ltd.
- BASF SE
- DuPont
- Mitsubishi Engineering-Plastics Corporation
- SABIC
- Celanese Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis