Sterile Compounding Pharmacies Market Size, Share, Growth and Forecast (2026 - 2036)
Sterile Compounding Pharmacies Market is segmented by Product (Sterile Injectable Compounding, Ophthalmic Sterile Preparations, Parenteral Nutrition (TPN), Sterile Hormone Preparations), Application (Hospital Pharmacy Services, Oncology Treatment Support, Ophthalmic Treatments, Pain Management Therapy), End Use (Hospitals, Specialty Clinics, Compounding Pharmacies, Ambulatory Surgical Centers), and Region, with forecasts covering the period from 2026 to 2036.
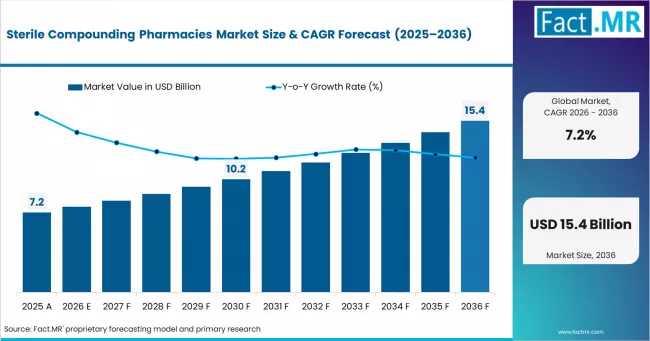
According to Fact.MR estimates, the global sterile compounding pharmacies market market was valued at USD 7.2 billion in 2025. The market is projected to reach USD 7.7 billion in 2026 and is expected to grow to USD 15.4 billion by 2036, expanding at a CAGR of 7.2%. Sterile Injectable Compounding is anticipated to account for 58.0% of the product segment in 2026, while Hospital Pharmacy Services is expected to remain the leading application with around 46.0% share.
Sterile Compounding Pharmacies Market Analysis and Forecast by Fact.MR
The global market for sterile compounding pharmacies is estimated to be around USD 7.7 billion in 2026 and has been forecast to expand at a CAGR of 7.2% to end up at USD 15.4 billion by 2036. The Sterile Compounding Pharmacies segment is led by Sterile Injectable Compounding, which is anticipated to account for 58.0% of the product segment in 2026, while Hospital Pharmacy Services is expected to remain the leading application with around 46.0% share. The market was valued at USD 7.2 billion in 2025 and is poised for steady growth driven by evolving industry dynamics and regional demand patterns.
Summary of Sterile Compounding Pharmacies Market
- Market Snapshot
- In 2025, the global Sterile Compounding Pharmacies Market was valued at approximately USD 7.2 billion.
- The market is estimated to reach USD 7.7 billion in 2026 and is projected to attain USD 15.4 billion by 2036
- The sterile compounding pharmacies market is likely to expand at a CAGR of 7.2% during the forecast period.
- The market is anticipated to create an absolute dollar opportunity of USD 7.7 billion between 2026 and 2036.
- Sterile Injectable Compounding account for 58.0% of market share in 2026.
- China (9.72%) and India (9.0%) are the key growth markets during the forecast period
- Demand and Growth Drivers
- Demand is shaped by the rising volume of complex therapies in oncology, pain management, and parenteral nutrition, where commercially available fixed-dose products cannot meet patient-specific dosing and stability requirements.
- Growth reflects stricter enforcement of USP 797 and USP 800 standards, which continues to push hospital systems toward outsourced 503B facilities that can deliver validated sterility compliance at scale.
- Adoption is increasing due to persistent drug shortages in hospital pharmacy procurement, with 503B outsourcers filling gaps for ready-to-administer injectables that in-house pharmacies cannot reliably produce.
- Product and Segment View
- Sterile injectable compounding leads the product segment with a 58% share in 2026, supported by rising oncology infusion volumes and a broader shift toward ready-to-administer formats.
- Hospital pharmacy services hold 46% of the application segment, reflecting the embedded role of in-hospital compounding units in acute care, ICU, and perioperative drug preparation.
- Hospitals account for 54% of end-use demand, with tertiary and academic medical centers driving the bulk of sterile preparation workloads tied to complex patient populations.
- Geography and Competitive Outlook
- North America remains the largest regional market, anchored by a mature 503B outsourcing ecosystem, strong FDA oversight, and concentrated demand from integrated hospital systems.
- Asia Pacific, led by China and India, is the fastest-growing region as tertiary hospital networks expand and oncology infusion volumes climb in line with rising cancer incidence.
- The competitive field is led by Fagron, Fresenius Kabi, PharMEDium Services, B. Braun, and Cantrell Drug, where scale in sterile filling, 503B registration, and hospital contracting define positioning.
- Analyst Opinion
- The sterile compounding pharmacies market is moving from a compliance-driven service layer into a strategic node of hospital drug supply. Growth is no longer tied only to shortage relief. It is being reshaped by oncology pipeline expansion, tighter USP 797 and 800 enforcement, and hospital interest in ready-to-administer formats that reduce bedside preparation risk. The decisive commercial lever is scale in 503B capacity combined with predictable regulatory performance, because hospital systems are consolidating their outsourcing base around fewer, audit-ready partners. Players that invest early in automated aseptic lines and multi-state 503B networks will capture disproportionate share of this incremental demand.
Why is Sterile Compounding Pharmacies Market Growing?
- Growth reflects the structural shift of injectable drug preparation away from bedside compounding toward centralized, validated facilities that reduce medication error and contamination risk.
- Demand is shaped by the expansion of oncology infusion centers, chronic disease treatment cycles, and hormone therapy volumes that require custom sterile dosing rather than fixed commercial formats.
- Adoption is increasing due to the migration of hospital systems toward outsourced 503B supply contracts that convert a clinical liability into a procurement decision.
The growth story sits at the intersection of regulation, safety, and drug access. USP 797 and USP 800 enforcement has redefined the minimum acceptable standard for in-hospital sterile preparation, and a meaningful share of smaller hospital pharmacies cannot economically meet those requirements. That creates a standing pipeline of outsourced demand flowing to registered 503B facilities with validated aseptic lines.
Oncology is the second engine. Customized cytotoxic drug preparation, biologic infusions, and supportive care injectables require patient-specific dose construction that commercial manufacturers cannot deliver. As cancer incidence rises in Asia Pacific and treatment protocols lengthen globally, infusion volumes feed directly into compounding throughput. Parenteral nutrition, ophthalmic formulations for intravitreal therapy, and hormone replacement pipelines add further demand layers.
The third driver is supply risk management. Persistent shortages of injectable drugs in the US and Europe have made hospital pharmacy leaders reliant on 503B partners to secure ready-to-administer alternatives. This reliance is sticky, because once a hospital validates an outsourcing relationship, switching costs are substantial.
Segment-wise Analysis of Sterile Compounding Pharmacies Market
- Sterile injectable compounding leads product demand with a 58% share in 2026, driven by oncology, ICU, and critical care dosing needs.
- Hospital pharmacy services dominate the application view at 46%, reflecting the tight integration between sterile compounding throughput and inpatient clinical workflows.
- Hospitals, with 54% end-use share, remain the core demand node, while 503B outsourcing facilities are the fastest-growing delivery model within the market.
The segmentation view reveals a concentrated market with well-defined commercial leverage points. Product, application, and end-use categories each cluster around one dominant sub-segment, which simplifies targeting but raises the bar on execution quality. Suppliers competing at the top of each category must defend share through regulatory reliability, throughput, and hospital-grade documentation rather than through product differentiation alone.
Why Does Sterile Injectable Compounding Lead the Product Segment?
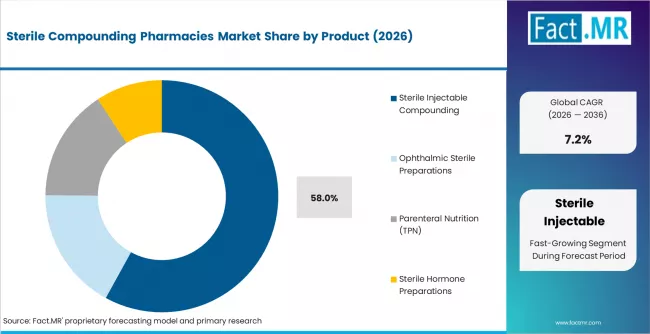
Sterile injectable compounding captures the highest share because it maps directly onto the most clinically sensitive use cases in modern hospital care. Oncology infusion, ICU drip dosing, post-surgical pain management, and parenteral nutrition all require sterile injectables prepared at patient-specific concentrations that commercial vials cannot cover. The segment also benefits from regulatory tailwinds, since USP 797 has pushed preparation out of satellite pharmacies and into validated facilities with stronger margin capture.
The leadership of this segment is structural rather than cyclical. Demand tracks therapy volumes, not compounding trends, which makes it relatively insulated from short-term procurement shifts. Over the forecast horizon, incremental growth will come from biologic infusions, expanded hormone replacement use, and custom ophthalmic intravitreal formulations.
Why Does Hospital Pharmacy Services Dominate the Application Segment?
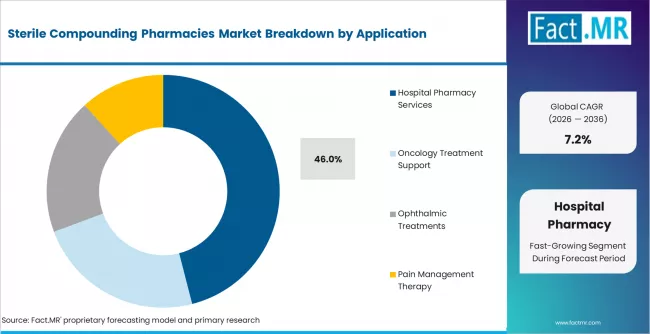
Hospital pharmacy services lead the application view because sterile compounding sits inside the daily drug preparation workflow of acute care facilities. ICU protocols, oncology infusion schedules, and perioperative dosing all depend on sterile preparations that must be available on demand, which ties compounding volume directly to hospital throughput. Even where hospitals outsource bulk sterile production to 503B facilities, final dose customization and just-in-time preparation remain in-house responsibilities.
Oncology treatment support is the fastest-growing application driver within the segment. Rising chemotherapy infusion volumes, biologic adoption, and the expansion of outpatient oncology centers translate into a durable demand base that does not depend on hospital in-patient throughput.
Key Growth Drivers, Constraints, and Market Scope
- Regulatory compliance under USP 797 and USP 800 is reshaping competitive dynamics in favor of scaled 503B operators.
- Oncology, parenteral nutrition, and ophthalmic therapy volumes are the three strongest demand anchors.
- Hospital consolidation is concentrating outsourcing contracts with a smaller set of qualified suppliers.
The commercial scope of the market is defined by three axes: regulatory risk, therapy demand, and procurement consolidation. Each axis rewards scale and penalizes fragmentation, which is why the competitive field is narrowing toward a smaller group of well-capitalized 503B facilities and integrated hospital compounding networks.
Growth Drivers
The primary driver is the clinical need for sterile, patient-specific drug preparation in oncology, ICU, parenteral nutrition, and ophthalmic therapy. This demand is non-substitutable, because commercial finished-dose products cannot meet the customization or stability requirements of these use cases. A secondary driver is sustained injectable drug shortage pressure across the US and Europe, which directly channels hospital demand into 503B outsourcing. A third driver is the aging patient base in developed markets and the expansion of tertiary care infrastructure in China, India, and Brazil, which lifts underlying sterile compounding volumes.
Constraints
The dominant constraint is regulatory exposure. Sterile compounding failures, contamination events, or FDA inspection findings can close facilities overnight and carry severe reputational consequences. This constraint caps the pace of capacity addition and keeps smaller, under-capitalized operators from scaling. A second constraint is labor availability, specifically trained compounding pharmacists and aseptic technique specialists, whose supply has not kept pace with demand. Workforce shortages limit how fast even well-funded 503B operators can expand throughput.
Opportunities
The strongest opportunity lies in oncology-focused outsourcing partnerships with integrated health systems, where ready-to-administer cytotoxic preparations reduce hospital risk and carry premium pricing. A second opportunity is automated compounding technology, where suppliers of robotic IV preparation systems and closed system drug transfer devices are capturing margin by selling to both hospital compounding units and 503B facilities. A third opportunity is the expansion of sterile ophthalmic and hormone compounding into ambulatory surgery and outpatient specialty clinics, which remain underpenetrated relative to hospital channels.
Regional Outlook Across Key Markets
.webp)
- China leads country-level growth at 9.72% CAGR, supported by tertiary hospital expansion, oncology infusion volume growth, and local 503B-equivalent facility build-out.
- India follows at 9.0%, driven by specialty hospital network expansion, rising cancer treatment access, and domestic sterile manufacturing scale-up.
- European demand is led by Germany at 8.28% and the UK at 6.12%, with both countries benefitting from tightening sterile preparation standards and oncology volume growth.
- North America remains the largest regional pool by absolute value, with the US at 6.84% and Canada supported by replacement demand and expanding 503B outsourcing contracts.
Country CAGR Table
| Country | CAGR (%) |
|---|---|
| China | 9.72% |
| India | 9.00% |
| Germany | 8.28% |
| Brazil | 7.56% |
| USA | 6.84% |
| UK | 6.12% |
| Japan | 5.40% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
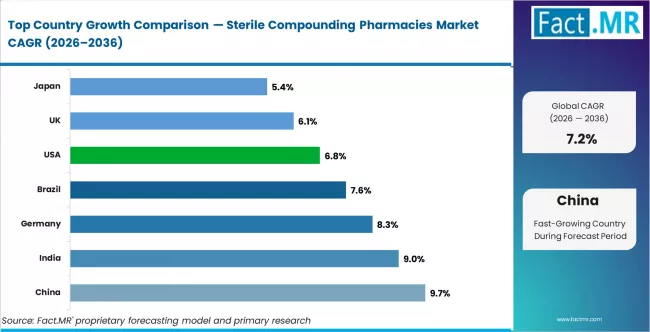
Country-level dynamics split cleanly into three clusters. Asia Pacific leads on volume growth as cancer incidence, hospital infrastructure spend, and sterile compounding capacity all climb together. Europe holds a mid-growth profile where regulatory tightening and oncology expansion offset slower baseline demand. North America, while slower in percentage terms, contributes the largest absolute-dollar opportunity because of its mature 503B outsourcing ecosystem and high per-hospital compounding spend.
United States
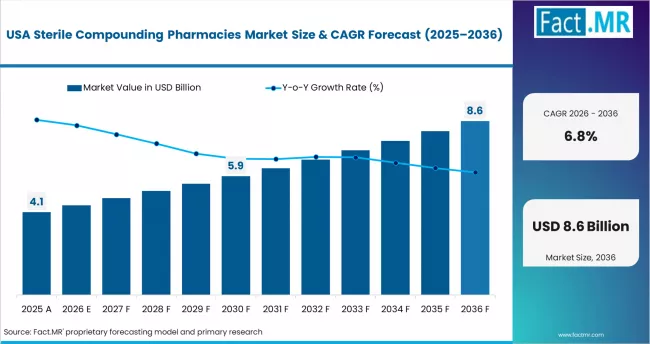
The United States anchors global demand for sterile compounding through its FDA-registered 503B facility network and its embedded hospital outsourcing culture. Growth is driven by oncology infusion volume expansion, persistent injectable drug shortages, and hospital preference for ready-to-administer doses that lower medication error risk. Large integrated delivery networks are consolidating compounding contracts with a narrower set of qualified suppliers, which favors scaled operators with multi-state reach.
- FDA 503B registration and inspection regime set the competitive bar for suppliers, rewarding scale and documentation depth.
- Oncology clinic expansion and hospital-at-home models are opening new distribution channels for compounded sterile therapies.
- Persistent shortages of injectable drugs create structural, non-cyclical demand for 503B outsourcing capacity.
Canada
Canada represents a tightly regulated sterile compounding market with demand concentrated in provincial hospital systems and specialty cancer centers. NAPRA guidelines and provincial college oversight shape sterile preparation standards, while cross-border supply relationships with US 503B providers fill capacity gaps. Growth is supported by oncology volumes, parenteral nutrition demand, and a gradual shift toward outsourced sterile preparation in mid-size hospitals.
- Provincial cancer agencies drive centralized oncology infusion contracts.
- NAPRA sterile compounding standards are progressively tightening.
- Cross-border US 503B sourcing is filling specialty sterile supply gaps.
United Kingdom
The United Kingdom sterile compounding market is shaped by NHS hospital pharmacy networks and a cluster of specialist aseptic units that serve regional oncology and parenteral nutrition demand. MHRA licensing and Section 10 exemptions define the regulatory envelope, with rising volumes tied to cancer treatment pathways and pediatric sterile formulations. The UK is also seeing gradual outsourcing of sterile preparation from smaller trusts to specialist aseptic hubs.
- NHS aseptic unit capacity constraints are driving outsourcing interest.
- MHRA oversight maintains high barriers to entry.
- Oncology and pediatric sterile preparation are the highest-growth pockets.
Germany
Germany leads European growth in sterile compounding on the back of well-established hospital pharmacy infrastructure, high per-capita oncology treatment rates, and a sizeable specialty compounding pharmacy segment. Federal pharmacy law and AMWHV regulations define preparation standards, while statutory health insurance reimbursement supports predictable volume flows. Growth is further lifted by parenteral nutrition demand and specialist hormone compounding services.
- AMWHV regulations sustain high-quality manufacturing standards.
- Statutory insurance reimbursement underpins stable demand volumes.
- Specialty compounding pharmacies capture premium outpatient sterile preparation demand.
China
China is the fastest-growing country in the forecast, driven by tertiary hospital network expansion, rising cancer incidence, and government-led investment in pharmacy preparation centers. Sterile compounding is moving from scattered in-hospital units toward centralized pharmacy intravenous admixture services (PIVAS), which operate at a scale comparable to 503B facilities in the US. This structural shift supports both demand expansion and consolidation.
- PIVAS deployment across tier-1 and tier-2 hospitals is accelerating centralized sterile preparation.
- Rising oncology incidence drives sustained infusion volume growth.
- Domestic sterile compounding capacity is scaling to reduce import dependency.
India
India is growing on the back of tertiary hospital chain expansion, rising oncology treatment access, and the deepening footprint of corporate hospital groups in metro cities. Sterile compounding is concentrated in large hospital systems and a small number of specialty compounding pharmacies. The CDSCO regulatory framework and hospital accreditation requirements are gradually raising quality standards across the segment.
- Corporate hospital chain expansion drives centralized sterile preparation demand.
- Oncology treatment access is broadening beyond metro markets.
- NABH accreditation requirements are tightening sterile preparation standards.
Competitive Benchmarking and Company Positioning
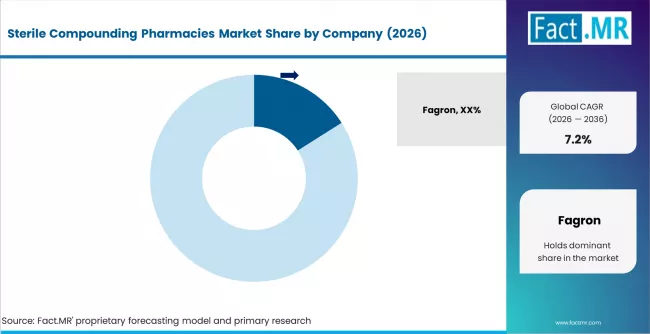
- Fagron leads with an estimated 16% share, anchored by its global compounding infrastructure and hospital-grade branded formulations.
- Fresenius Kabi and B. Braun compete on the basis of integrated sterile injectable capacity and hospital contracting depth.
- PharMEDium Services and Cantrell Drug hold differentiated 503B positions in the US market, with specialization in ready-to-administer hospital supply.
The competitive landscape is moderately concentrated, with the top five players holding a combined share that reflects the scale advantage of regulated 503B operations. Fagron's lead is reinforced by its proprietary branded compounding lines and its ability to serve both hospital and specialty compounding pharmacy channels globally. Fresenius Kabi and B. Braun bring the weight of integrated sterile injectable manufacturing, which gives them structural cost and supply advantages in hospital tenders.
Competition sits less on product innovation and more on regulatory reliability, capacity depth, and hospital contracting relationships. 503B facility operators that have avoided major FDA findings and have multi-state reach command pricing premiums. The second tier of the market, occupied by specialist pharmacies and outsourced facilities, competes on niche therapy areas such as ophthalmic, hormone, and 503A patient-specific services.
Strategic priorities across the field include expanding ready-to-administer portfolios for hospital oncology and ICU channels, investing in robotic and gravimetric compounding systems that reduce human error, and building multi-site networks to absorb hospital consolidation. Players that combine strong regulatory track records with scaled automation will capture disproportionate incremental demand over the forecast period.
Competitive Analysis
The competitive edge in this market is built around three compounding dimensions: regulatory reliability, automation, and hospital contracting depth. Fagron's position reflects all three, combining branded compounding lines with strong pharmacy channel presence. Fresenius Kabi and B. Braun leverage parent-company injectable manufacturing scale to protect margins on high-volume hospital formats. PharMEDium Services plays deep in US 503B outsourcing, focusing on ready-to-administer hospital supply. Cantrell Drug and Institutional Pharmacy Solutions serve niche 503B pockets with specialized therapy portfolios.
Second-tier competitors maintain relevance by focusing on specific therapy areas, such as sterile hormone preparations, ophthalmic formulations, or pain management dosing. Smaller 503A pharmacies remain valuable to localized physician networks but lack the capacity or documentation to scale into hospital system contracts. The gap between scaled 503B operators and smaller pharmacies continues to widen as regulatory enforcement intensifies.
Benchmarking Table
| Company | Scale Reach | Regulatory Track Record | Automation Depth | Therapy Portfolio |
|---|---|---|---|---|
| Fagron | Global | Strong | High | Broad |
| Fresenius Kabi AG | Global | Strong | High | Broad |
| B. Braun Melsungen AG | Global | Strong | Medium-High | Broad |
| PharMEDium Services LLC | United States | Strong | High | Ready-to-Administer Focus |
| Cantrell Drug Company | United States | Strong | Medium | 503B Specialty |
| Institutional Pharmacy Solutions | United States | Medium | Medium | Niche Hospital Supply |
Recent Developments
- In 2025, Fagron continued the expansion of its US 503B compounding footprint, strengthening its ready-to-administer hospital supply portfolio and deepening its presence with integrated delivery networks.
- In 2025, Fresenius Kabi advanced its compounding automation investments, expanding capacity for oncology and parenteral nutrition preparation across US and European sites.
- In 2026, B. Braun reinforced its sterile injectable and compounding services footprint through additional hospital contracting in North America and Europe.
Leading Companies Shaping Sterile Compounding Pharmacies Market
-
Major Players
- Fagron
- Fresenius Kabi AG
- PharMEDium Services LLC
- B. Braun Melsungen AG
- Cantrell Drug Company
- Institutional Pharmacy Solutions
- Lorraine's Pharmacy
-
Emerging Players
- QuVa Pharma
- Avella Specialty Pharmacy
- Central Admixture Pharmacy Services (CAPS)
- Empower Pharmacy
- Leiter's Health
- Wedgewood Pharmacy
- Olympia Pharmacy
- Imprimis Pharmaceuticals
Sources and Research References
- United States Food and Drug Administration (FDA) - 503B Outsourcing Facility Registration and Inspection Reports
- United States Pharmacopeia (USP) - USP 797 and USP 800 Chapter Revisions
- World Health Organization (WHO) - Global Cancer Observatory and Medicines Access Reports
- National Association of Boards of Pharmacy (NABP) - Compounding Pharmacy Accreditation Standards
- Fresenius Kabi AG - Annual Report and Sterile Injectable Operations Disclosures
- Fagron - Investor Reports and Compounding Network Updates
- Primary interviews with hospital pharmacy directors, 503B facility operators, and sterile compounding equipment suppliers
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the current and future size of the sterile compounding pharmacies market?
- How fast is the market expected to grow between 2026 and 2036?
- Which product segment is likely to lead the market by 2026?
- Which application segment will account for the highest demand by 2026?
- What factors are driving demand for sterile compounding globally?
- How is the shift toward 503B outsourcing reshaping hospital procurement?
- Why are China and India emerging as the fastest-growing markets?
- Which countries are projected to show the fastest growth through 2036?
- Who are the key companies active in the sterile compounding pharmacies market?
Sterile Compounding Pharmacies Market Definition
The sterile compounding pharmacies market covers facilities and services engaged in the preparation of patient-specific or batch-level sterile drug products under controlled aseptic conditions. This includes injectable, ophthalmic, and infusion-grade formulations prepared in hospital-based compounding units, 503A pharmacies serving individual patient prescriptions, and 503B outsourcing facilities supplying office-use doses to healthcare providers.
Sterile Compounding Pharmacies Market Inclusions
The scope includes sterile injectable compounding, IV infusion preparations, ophthalmic sterile products, parenteral nutrition, sterile hormone preparations, and pain management formulations. It captures value across product type, application, end use, technology, packaging, and distribution channel. Coverage spans hospital-owned compounding units, 503A retail pharmacies, 503B outsourcing facilities, specialty compounding pharmacies, and integrated hospital network contracts from 2026 to 2036.
Sterile Compounding Pharmacies Market Exclusions
The scope excludes non-sterile compounded preparations, commercially manufactured finished-dose pharmaceuticals sold under NDA or ANDA pathways, and over-the-counter formulations. It also excludes raw API manufacturing, contract drug development and manufacturing (CDMO) activity unrelated to sterile compounding, veterinary-only compounding without human use crossover, and non-pharmacy sterile medical device preparation services.
Sterile Compounding Pharmacies Market Research Methodology
- Primary Research: Interviews with hospital pharmacy directors, 503B facility operators, sterile compounding equipment vendors, and procurement leads across integrated delivery networks to validate demand patterns, pricing structures, and contract dynamics.
- Desk Research: Review of FDA 503B facility registrations, USP 797 and USP 800 guidance revisions, hospital system outsourcing tenders, and published financial disclosures of listed compounding service providers.
- Market Sizing: Demand-side model built on therapy volumes (oncology, pain, parenteral nutrition, ophthalmic), compounded dose unit economics, and regional compounding penetration benchmarks, cross-checked with top-down estimates.
- Data Validation: Triangulation across primary inputs, regulatory filings, and hospital procurement data, with sensitivity checks on oncology volume growth and 503B capacity expansion trajectories.
Scope of Analysis
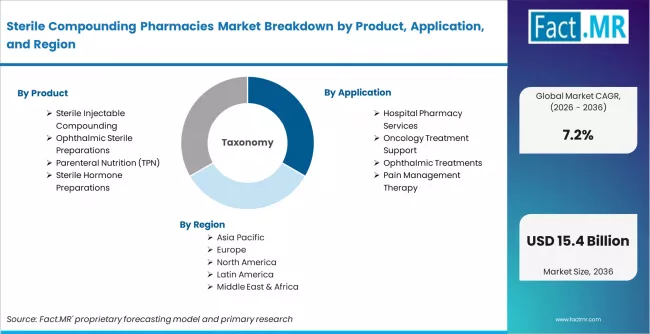
| Parameter | Details |
|---|---|
| Quantitative Units | USD 7.69 billion to USD 15.42 billion, at a CAGR of 7.2% |
| Market Definition | The sterile compounding pharmacies market covers patient-specific and batch-level sterile drug preparation across 503A, 503B, and hospital pharmacy compounding channels, including injectable, ophthalmic, infusion, and hormone formulations. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, Canada, UK, Germany, China, India, Brazil, Japan, 30 plus countries |
| Key Companies | Fagron, Fresenius Kabi AG, PharMEDium Services LLC, B. Braun Melsungen AG, Cantrell Drug Company, Institutional Pharmacy Solutions, Lorraine's Pharmacy |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on therapy-level volume inputs, 503B capacity benchmarking, hospital contracting pricing analysis, and primary interviews across hospital pharmacy leaders, 503B operators, and specialty compounding pharmacies |
Market Segmentation Analysis
-
Sterile Compounding Pharmacies Market Market Segmented by Product:
- Sterile Injectable Compounding
- IV Infusion Preparations
- Patient-Specific Sterile Injections
- IV Infusion Preparations
- Ophthalmic Sterile Preparations
- Eye Drops & Intravitreal Solutions
- Post-Surgical Ophthalmic Formulations
- Eye Drops & Intravitreal Solutions
- Parenteral Nutrition (TPN)
- IV Nutritional Formulations
- Hospital Critical Care Nutrition
- IV Nutritional Formulations
- Sterile Hormone Preparations
- Injectable Hormone Therapy
- Personalized Endocrine Medications
- Injectable Hormone Therapy
- Sterile Injectable Compounding
-
Sterile Compounding Pharmacies Market Market Segmented by Application:
- Hospital Pharmacy Services
- Inpatient Sterile Medication Preparation
- ICU & Surgical Care Compounding
- Inpatient Sterile Medication Preparation
- Oncology Treatment Support
- Cancer Infusion Therapies
- Chemotherapy Dose Customization
- Cancer Infusion Therapies
- Ophthalmic Treatments
- Eye Surgery Medication Support
- Intravitreal Drug Delivery
- Eye Surgery Medication Support
- Pain Management Therapy
- Customized Injectable Pain Drugs
- Chronic Pain Treatment Solutions
- Customized Injectable Pain Drugs
- Hospital Pharmacy Services
-
Sterile Compounding Pharmacies Market Market Segmented by End Use:
- Hospitals
- Tertiary Care Hospitals
- Surgical & ICU Units
- Tertiary Care Hospitals
- Specialty Clinics
- Oncology & Ophthalmology Clinics
- Pain Management Centers
- Oncology & Ophthalmology Clinics
- Compounding Pharmacies
- Outsourced 503A Facilities
- 503B Outsourcing Facilities
- Outsourced 503A Facilities
- Ambulatory Surgical Centers
- Day Care Surgery Units
- Outpatient Infusion Centers
- Day Care Surgery Units
- Hospitals
-
Sterile Compounding Pharmacies Market Market Segmented by Technology:
- Aseptic Compounding Technology
- ISO Class Cleanroom Systems
- Sterile Barrier Isolation Systems
- ISO Class Cleanroom Systems
- Automated Compounding Systems
- Robotic IV Preparation Systems
- Closed System Drug Transfer Devices
- Robotic IV Preparation Systems
- Sterile Filtration Technology
- Microbial Filtration Systems
- Endotoxin Removal Systems
- Microbial Filtration Systems
- Gravimetric Dispensing Systems
- Precision Dose Measurement Systems
- Digital Compounding Controls
- Precision Dose Measurement Systems
- Aseptic Compounding Technology
-
Sterile Compounding Pharmacies Market Market Segmented by Packaging:
- Sterile IV Bags & Vials
- Pre-Filled Syringes
- Sterile Injectable Containers
- Pre-Filled Syringes
- Compounding Kits
- Single-Use Sterile Kits
- Hospital Dose Preparation Packs
- Single-Use Sterile Kits
- Ampoules & Bottles
- Sealed Sterile Drug Containers
- Light-Protected Packaging Systems
- Sealed Sterile Drug Containers
- Sterile IV Bags & Vials
-
Sterile Compounding Pharmacies Market Market Segmented by Distribution Channel:
- Hospital Pharmacies
- In-Hospital Compounding Units
- Clinical Pharmacy Networks
- In-Hospital Compounding Units
- Specialty Pharmacies
- Sterile Compounding Providers
- Outsourced 503B Facilities
- Sterile Compounding Providers
- Online Medical Supply
- B2B Pharmaceutical Platforms
- Healthcare Procurement Portals
- B2B Pharmaceutical Platforms
- Hospital Pharmacies
-
Sterile Compounding Pharmacies Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the sterile compounding pharmacies market in 2026?
The global sterile compounding pharmacies market is estimated to be valued at USD 7.69 billion in 2026.
What will be the size of the sterile compounding pharmacies market in 2036?
The market size is projected to reach USD 15.42 billion by 2036.
How much will the sterile compounding pharmacies market grow between 2026 and 2036?
The market is expected to grow at a 7.2% CAGR between 2026 and 2036.
Which product segment is expected to lead the market in 2026?
Sterile injectable compounding is expected to lead the product segment with a 58% share in 2026.
Which country is the fastest-growing market?
China is the fastest-growing country in the forecast, projected to grow at a 9.72% CAGR through 2036.
Why is India a key growth market?
India is projected to grow at a 9.0% CAGR, driven by hospital network expansion, oncology access, and rising sterile preparation standards.
What does the sterile compounding pharmacies market cover?
The market covers patient-specific and batch-level sterile drug preparation across 503A, 503B, and hospital pharmacy channels, including injectable, ophthalmic, infusion, and hormone formulations.
How is the market forecast estimated and validated?
The forecast is built on a hybrid demand-side and top-down model using therapy volumes, 503B capacity benchmarks, hospital contracting data, and primary interviews across the compounding value chain.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Sterile Injectable Compounding
- IV Infusion Preparations
- _Patient-Specific Sterile Injections
- Ophthalmic Sterile Preparations
- Eye Drops & Intravitreal Solutions
- _Post-Surgical Ophthalmic Formulations
- Parenteral Nutrition (TPN)
- IV Nutritional Formulations
- _Hospital Critical Care Nutrition
- Sterile Hormone Preparations
- Injectable Hormone Therapy
- _Personalized Endocrine Medications
- Sterile Injectable Compounding
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Hospital Pharmacy Services
- Inpatient Sterile Medication Preparation
- _ICU & Surgical Care Compounding
- Oncology Treatment Support
- Cancer Infusion Therapies
- _Chemotherapy Dose Customization
- Ophthalmic Treatments
- Eye Surgery Medication Support
- _Intravitreal Drug Delivery
- Pain Management Therapy
- Customized Injectable Pain Drugs
- _Chronic Pain Treatment Solutions
- Hospital Pharmacy Services
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Tertiary Care Hospitals
- _Surgical & ICU Value (USD Million)s
- Specialty Clinics
- Oncology & Ophthalmology Clinics
- _Pain Management Centers
- Compounding Pharmacies
- Outsourced 503A Facilities
- _503B Outsourcing Facilities
- Ambulatory Surgical Centers
- Day Care Surgery Value (USD Million)s
- _Outpatient Infusion Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Aseptic Compounding Technology
- ISO Class Cleanroom Systems
- _Sterile Barrier Isolation Systems
- Automated Compounding Systems
- Robotic IV Preparation Systems
- _Closed System Drug Transfer Devices
- Sterile Filtration Technology
- Microbial Filtration Systems
- _Endotoxin Removal Systems
- Gravimetric Dispensing Systems
- Precision Dose Measurement Systems
- _Digital Compounding Controls
- Aseptic Compounding Technology
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Sterile IV Bags & Vials
- Pre-Filled Syringes
- _Sterile Injectable Containers
- Compounding Kits
- Single-Use Sterile Kits
- _Hospital Dose Preparation Packs
- Ampoules & Bottles
- Sealed Sterile Drug Containers
- _Light-Protected Packaging Systems
- Sterile IV Bags & Vials
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacies
- In-Hospital Compounding Value (USD Million)s
- _Clinical Pharmacy Networks
- Specialty Pharmacies
- Sterile Compounding Providers
- _Outsourced 503B Facilities
- Online Medical Supply
- B2B Pharmaceutical Platforms
- _Healthcare Procurement Portals
- Hospital Pharmacies
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By End Use
- By Technology
- By Packaging
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Fagron
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Fresenius Kabi AG
- PharMEDium Services LLC
- Institutional Pharmacy Solutions
- Cantrell Drug Company
- Lorraine’s Pharmacy
- B. Braun Melsungen AG Company
- Fagron
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Packaging
- Figure 18: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Distribution Channel
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Packaging
- Figure 47: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Distribution Channel
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Packaging
- Figure 66: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 78: Western Europe Market Attractiveness Analysis by End Use
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Packaging
- Figure 85: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 116: East Asia Market Attractiveness Analysis by End Use
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 122: East Asia Market Attractiveness Analysis by Packaging
- Figure 123: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis